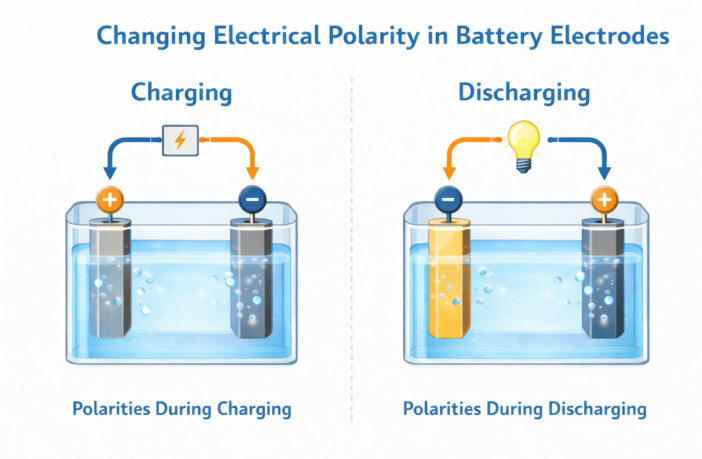
All batteries have two electrodes connected to external terminals. One of these is the anode, and the other is the cathode. We investigate changing electrical polarity in batteries, as they transition between charging and discharging. Essentially, the positive one becomes the negative one, and vice-versa.
Electrode Mode in Discharging Batteries
The anode electrode is negative while a battery is discharging to a device, while the cathode has positive polarity. The anode may be graphite, although there are many exceptions.
The cathode, on the other hand, is usually an oxide of the active metal. This could, for example, be a zinc-metal oxide, a lithium-metal oxide, an aluminum-metal oxide, and so on.
We release electrons from the active metal atoms at the anode when we discharge a battery to a device. This causes the atom remnants to become metal ions:
- These ions travel through the electrolyte in the battery to reach the cathode.
- Electrons can’t travel through electrolyte, so they move through the external circuit.
- This external flow of electrons is the electrical current that powers that device.
The ions and electrons reach the battery cathode, which accepts them.The cathode is positive relative to the anode during discharge. This is because of the electrochemical potential difference between the electrodes, during the first phase of changing electrical polarity in batteries.
Reversing Changed Electrode Polarity in Batteries
The process we described reverses, when we recharge a rechargeable battery. The power from the battery charger sends the electrons and ions back to the anode, restoring the battery to its original polarity.
The names ‘anode’ and ‘cathode’ thus reflect the direction of the chemical reactions, rather than simply confirming the positive or negative sign.
This is why, in rechargeable batteries, the polarity of the electrodes changes during charging. However, during normal battery use, the anode is still negative and the cathode is still positive.
More Information