Gaston Planté could hardly have imagined mass-recycling spent batteries, when he invented lead-acid chemistry back in 1859. Just getting the thing to work was his sole interest. Since then, the battery industry has focused on developing more powerful batteries. Recycling their materials was someone else’s problem, it seemed.
Well that was true until researchers at Massachusetts Institute of Technology (MIT) came up with a better way. That better way is a battery that falls apart on cue.
A Better Battery That Falls Apart on Cue
The MIT researchers turned the battery recycling paradigm on its head. We already have batteries that perform well, they decided. Well, that is until we try to take them apart for recycling. That’s the moment the problem starts.
What if we build a battery that falls apart on cue instead, they pondered. Once we have that right, they continue, then we could use our collective knowledge to improve its performance. We just had to share this ‘upside down’ type of thinking with our followers!
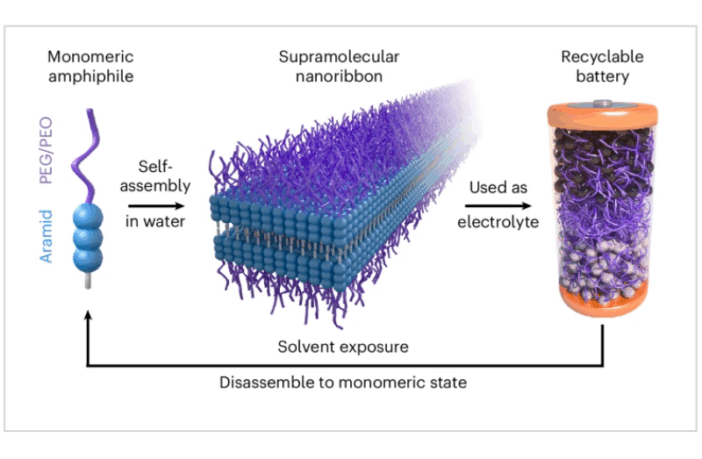
After we imagine a fair amount of to-ing and fro-ing, the MIT researchers developed a new type of self-assembling material, according to Innovation News Network. In this instance the new material was a novel electrolyte for a lithium-ion battery.
The Novel Electrolyte That Dissolves Itself
The research report in Springer Nature that we link to below, explains how this novel electrolyte material is able to rapidly dissolve. Once it does so, the rest of the battery literally falls apart. A battery recycler can then separate out the other individual components, and recycle them.
This discovery at Massachusetts Institute of Technology has potential to spin out into greater efficiency when it comes to recycling electric vehicle batteries. It could conceivably help secure a new local supply chain, comprising recycled cobalt, nickel, and lithium minerals.
More Information
Safer Recycling of Lithium-Ion Batteries
Why Fuss Over Recycling Lithium Ions Now?
Preview Image: Disassembling a Battery