Hydrogen fuel cells have proved almost prohibitively expensive on account of their manufacturing cost, and very low net energy value. Alkaline fuel cells fare better because of their cheaper and more versatile hydrocarbon-based membranes. We introduce the topic, and browse the history before providing an overview of the chemistry.
A Brief History of Alkaline Fuel Cells
Francis Thomas Bacon developed the first alkaline fuel cells in 1939, using potassium hydroxide as electrolyte. He teamed this with porous “gas-diffusion” electrodes, instead of the acid electrolytes and solid electrodes in previous fuel cell prototypes.
Bacon also hit on the idea of using gases under pressure to resist the electrolyte flooding the electrodes. He continued experimenting through to the early 1960’s, when Pratt & Whitney aerospace manufacturers licensed the patents. They subsequently won the contract to power Apollo spacecraft with alkaline cells.
Chemical Reactions Inside The Cells
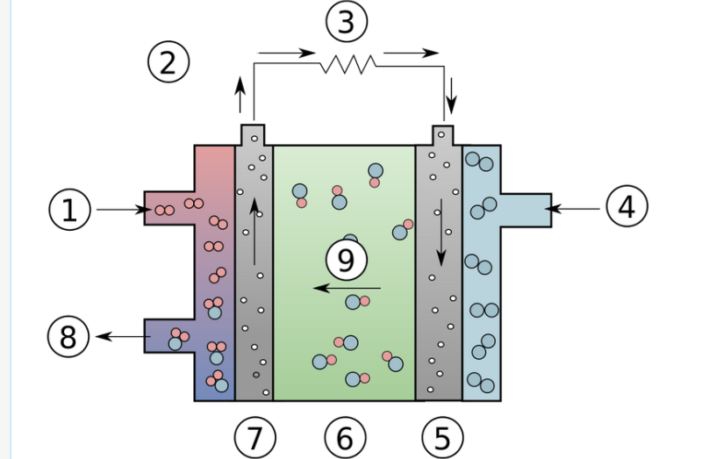
Depending on the details of their design, the operating temperature ranges from room temperature all the way up to 482°F / 250°C. Power-generating efficiencies may be as high as 70%, Colleen Spiegel assures us writing in Fuel Cell Store. However, in all cases:
- The oxygen reacts at the cathode to produce one of two types of ion.
- These may be hydroxide, or carbonate ions, depending on the electrolyte.
- The ions travel through the electrolyte to react with the hydrogen.
- Meanwhile, electrons flow through an external device powering it.
The electrodes may be either hydrophobic (water-resistant) or hydrophilic (attracted to water). The former are carbon-based, while the latter are metallic nickel, or nickel derivative. Precious-metal catalysts are usually platinum, or platinum alloys. However, the non-precious ones may be high-active nickel at the anode, and silver-based powders at the cathode.
The electrolyte has traditionally been an aqueous solution of alkaline potassium hydroxide, soaked in a matrix in mobile or immobile form. This alkaline environment permits the use of non-precious metals, considerably lowering the cost per unit.
More Information
Hydrogen Fuel Cell Basics How They Work
Better Solid State Batteries with New Pathways
Preview Image: Schematic of Alkaline Fuel Cell
Source Article by Dr. Colleen Spiegel