Sometimes it pays to take a step back, and review the basics behind the solid, dependable lead-acid batteries we supply. For if we do not, then we may forget the remarkable chemical reaction that takes place inside their handsome red cases. Without further ado here is what you need to know all about batteries.
All About Batteries in the Beginning
Commercial batteries have stored chemical energy successfully for well over a century. Our lead-acid ones follow the same principles Gaston Planté developed in 1859. This means they are able to power a variety of devices until it’s time for a recharge.
Basic Components in Every Battery
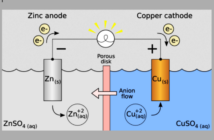
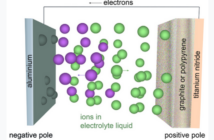
We’ll continue sharing all about batteries as we move on to the components. There are actually only three, beside the case containing them. How many other electrical devices do you know that are so simple? Briefly, there are two metal electrodes per cell of different types, and an electrolyte that regulates how they perform.
One electrode is more reactive than the other. This means negatively charged ions can flow from it to the less reactive one, while electricity travels through a device between the terminals. When all the negatively charged ions have relocated the battery is ‘flat’. Recharging the battery – if the design allows it – returns them to where they came from.
Bring This Theory to Life with a Lemon
We use diluted sulfuric acid as ‘gatekeeper’ electrolyte in our lead acid batteries. A lemon contains a decent amount of citric acid. We could light up an LED if we had suitable electrodes. In this case a copper-plated zinc penny and a galvanized zinc nail are all we need.
It’s amazing when we see that happen before our very eyes! Could batteries be that simple? Yep they really are, although in practice battery design is not that straightforward. But that’s another story for another day, as the saying goes.
Breaking News
Battery Passport to Trace Content and Carbon
What Happens If You Can’t Get an EV Battery?