Aqueous batteries have been in the news lately, with fascinating research findings that we share here. We decided it was time to write a basic consumer guide to these aqueous batteries, to tie the loose threads together. Aqueous batteries have been around for a long time, ever since Gaston Planté invented the lead-acid battery in 1859, in fact.
Aqueous Batteries Are Safer and More Reliable
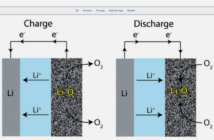
Aqueous batteries use an electrolyte which contains water, and therefore cannot catch fire. Lead acid battery electrolyte is sulfuric acid diluted with distilled water, although there are other options available nowadays.
Alkaline aqueous batteries appeared at the beginning of the 20th century, with a potassium hydroxide electrolyte containing water. Nickel-metal hydride batteries appeared in the 1980s. By 2020, rechargeable aqueous batteries controlled half the global market.
Wikipedia confirms that aqueous batteries are generally safer, more reliable, and relatively inexpensive in comparison to lithium-ion. Although lithium-ion batteries dominate the market because they are more powerful.
Pros and Cons of Aqueous for Consumers
Why Aqueous Batteries Are Still Popular
Batteries with water in their electrolytes are safer, because they cannot burn. They are also robust. They can tolerate mishandling, and overcharging thanks to oxygen gas recombining with the water in the electrolyte.
Aqueous batteries are also cheaper to manufacture. Their electrolyte components are less expensive than lithium-ion battery alternatives. Moreover, their inherent safety counters the need for electronic safeguards.
Downside Of Aqueous Chemistry For Consumers
Our consumer guide to aqueous batteries would be incomplete, if we stopped our review at this point. The water in the electrolyte begins electrolizing at 1.23 volts potential, compared to lithium-ion at 3.3 to 3.9 volts. Although smart technology may stretch aqueous batteries to 2.0 volts.
This difference makes lithium-ion batteries 2 – 3 times more powerful, in terms of volume and weight density. We also have to mark aqueous batteries down in terms of cycle life, and the tendency of the water to corrode the components.
There is renewed interest in the chemistry we review in this consumer guide to aqueous batteries. There is a counter movement against the environmentally unfriendly nature of unstable lithium-ion batteries. Come back soon to discover more developments in our fascinating world of batteries.
More Information
Early Days of Lead-Acid Battery History