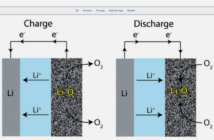
We touched on how batteries work in our previous post. You may recall how a battery sacrifices its chemical energy in favor of the electrical energy it sends through an external circuit. This creates an imbalance which it corrects by sending cations and ions to the cathode and anode respectively. We continue our discussion by outlining the different battery categories.
How The Four Main Battery Categories are Different
The Materials Science and Energy Education Department of Washington University highlights four different battery categories as follows:
- Affordable galvanic primary batteries that we can only use once.
- More expensive secondary batteries we can use a number of times.
- Fuel cells that can theoretically work for as long as fuel is available.
- Reserve batteries where a part remains isolated until the battery activates.
We have written quite extensively about the first three battery types. We dedicate the rest of this post to briefly describe the working principles of reserve batteries.
The Four Main Types of Reserve Batteries
Reserve batteries, or standby ones, are primary, single-use cells or sets. Their active components are kept separate to prevent gradual self-discharge while in storage. This makes them ideal for emergency equipment that may remain idle for years.
Dry cell auto starter batteries, where dealers add electrolyte are another good example of the technology in action. Other common examples include:
- Zinc-air and aluminum-air batteries, where a user removes a tab over the air intake.
- Thermal batteries that remain inactive until an external heat source melts their electrolyte.
- Water-activated batteries, where users add water allowing electrons to flow between terminals.
Applications for These Different Battery Categories
Citizens may be more likely to encounter reserve batteries in hearing aids, where non-toxic zinc-air batteries are popular. Reserve batteries in buoys activate automatically in ocean water.
However because of cost, reserve batteries primarily power military projectiles where acceleration activates them. Other weapons use small containers of pressurized air to force the electrolyte from a storage tank into a battery.
More Information
Could Zinc-Air Batteries Beat Lithium-Ion?
Preview Image: Zinc-Air Hearing Aid Batteries