We continue our journey into the world of electro-chemistry, where scientists now observe events through electron microscopes. The founders of our science could however, only have imagined what was happening, as they sometimes endangered their lives. Why do we need an electrolyte, they may have pondered. What is the role of electrolytes in electro-chemistry?
Fundamentals Governing Electrolytes in Electro-Chemistry
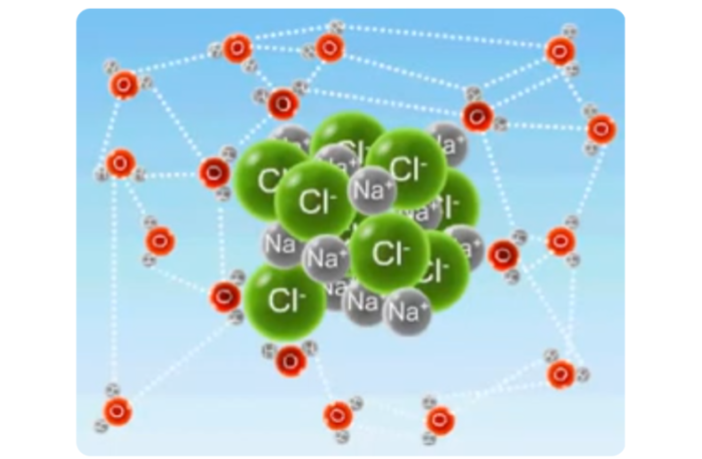
Britannica explains how the active properties of an electrolyte depend on electrically-charged atoms being present. These microscopic substances may be innate, or the result of groups of atoms appearing after spontaneous splitting up, or dissociation of their molecules. The intensity of their presence varies, because:
- Strong electrolytes have already dissociated some or perhaps all of their molecules into charged particles, or ions.
- Whereas weaker electrolytes have not completed the process, so fewer charged particles, or ions are present.
However, there must be a difference in electrical potential between the electrodes, for these ions to flow across to them. They must also be in physical contact with an electrolyte to make this possible. Under these two conditions:
- Positive charges will move from the electrolyte towards the cathode.
- Ions with negative charges will conversely move towards the anode.
As ions reach their destination, they donate their charge to the metal, or receive a charge. The ions then become ordinary neutral atoms, or groups of atoms according to Britannica, resulting in a chemical change.
A Brief Introduction to Redox Reductions
Redox is an abbreviation for ‘reduction-oxidation’, affecting electrolytes in the chemistry of batteries. This process removes electrons from one electrode, thereby reducing its supply.
Or alternatively it gives electrons to another electrode, thereby increasing its own supply in a process we call oxidization. Electrodes affected this way are either ‘reduced’ or ‘oxidized’ depending on their state at the time. Visit our next post to learn more about this amazing branch of science.
More Information
Solid–Electrolyte Interphase – Dramatic News
Electro-Chemical Reactions Unpacked For You
Preview Image: Dissolving Electrolytes in Water
YouTube Share Link: https://youtu.be/kkSOS_Z1mQM?si=ts1J_FGS8DsGDJZ7