Lithium-ion batteries may burn when they overheat, because their electrolyte is flammable and can catch fire. Non-flammable aqueous electrolytes cannot do so, because their main constituent is water, and water suppresses fires. This is why lead-acid electrolyte cannot ignite in our batteries. But how is this possible when water (H2O) contains flammable hydrogen, and oxygen that supports combustion?
Why Can’t the Lead-Acid Electrolyte in Our Batteries Catch Fire?
Science ABC explains this enigma as follows. Water does not burn because, “it’s already been through the process of combustion”. But I thought you said, “water cannot burn” we hear you say. Let’s drill down deeper into the riddle, shall we.
Water is a composition of hydrogen and oxygen atoms. Both of these support combustion, and so therefore water should burn. But why then does water extinguish fires, when logic suggests it should encourage them? Is this why lead-acid electrolyte cannot ignite either? After all, it is a 75% dilution of sulfuric acid and water.
Water forms when we burn hydrogen. Therefore scientists assure us water does not burn, because it has burned already. That opens the next question in our minds. What happens inside a fire when it burns?
What Goes On Inside a (Hydrogen) Fire?
Hydrogen burning in air reacts with the oxygen to form water with the simultaneous release of energy. This is a special case of any fire, which in essence combines two molecules and atoms in order to release energy as light and heat.
To start any fire we require these three things:
- A combustible material like paper, wood, hydrogen, etc.
- An oxidizer of which oxygen is commonest in our world.
- A source of heat to kick start the combustion process.
Water is a wonderful medium for smothering flames and robbing them of oxygen. Next time somebody tells you the chemical formula for water is H20, you can answer I know that, and that’s why it cannot burn.
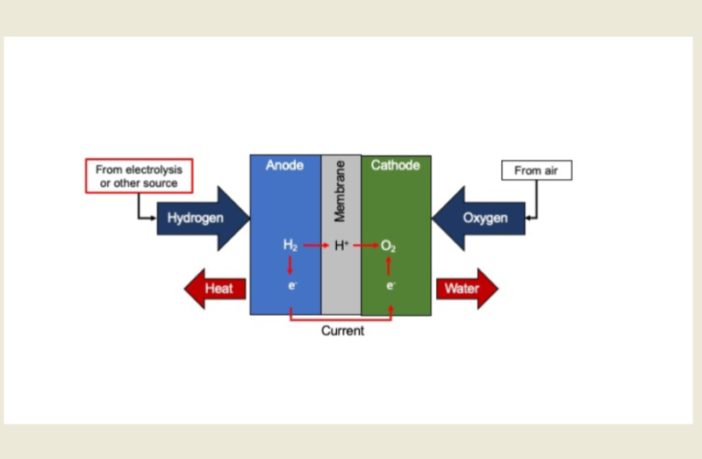
More Information
Hydrogen Fuel Cell Basics How They Work