An electron is a stable, sub-atomic particle found in all atoms, and with a charge of negative electricity. A battery works by exchanging electrons between two chemical reactions, namely oxidation and reduction. The former is the loss of electrons, while the latter is a corresponding gain. This is how a battery works in broad terms, but there is more to know.
How a Battery Energizes An Electrical Load
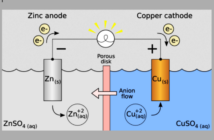
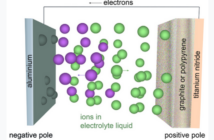
An electrical load is any part of a circuit that consumes electrical energy. The energy in a battery can power an external load, by isolating the oxidation from the reduction reaction. This flow then passes through the load between the two battery terminals instead. Meanwhile, ions flow in the opposite direction between the electrodes to compensate for this process.

The main components of an electro-chemical battery in the diagram above are a pair of electrodes, an electrolyte, and a separator. The oxidations and reductions occur in the electrodes, while these in turn connect to the terminals and the external load.
The polarity of the electrodes depends on whether the battery is charging or discharging. For example, when it is discharging:
- The oxidation reaction occurs at the anode which has a positive charge.
- While the reduction reaction occurs at the cathode. which has a negative charge.
More About The Balancing Flow Of Ions
When electricity flows from one terminal to another through an external load, this affects the balance between the anode releasing it, and the cathode receiving it. Ions flow in the opposite direction through the electrolyte to compensate for this process, and prevent a charge build-up according to PV Education website.

That was a very basic description of how a battery works by way of an introduction. We’ll be following up by explaining how voltage, current and general battery performance depend on the materials in different batteries.
More Information
Anode and Cathode Making a Perfect Pair