Did you ever wonder how a battery lights a torch bulb, starts an auto or fires up a laptop? The secret is in the chemistry. So let’s get going right away, and explore how batteries work. All batteries follow the same principle. They store electrical energy – not electricity – in the chemicals inside their case. Then they release it when we connect their terminals together. This should be through a device that controls the flow, to avoid a short circuit.
A Battery Comprises Three Main Components

Firstly, there are two terminals, namely the connecting points. A terminal is a starting or an end point, in this case the beginning and the end of the electrical energy flow. We call these terminals the cathode and the anode.
The cathode has the plus sign next to it making it the positive. The negative anode may have a minus sign. It is as simple as that!
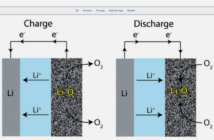
The cathode and the anode comprise metallic chemicals able to exchange electrons. We separate them with an electrolyte that controls the rate of flow. When we connect the device, the energy flows through it from the negative to the positive terminal. Thus, that’s how a battery lights a torch bulb, starts an auto or fires up a laptop.
How a Battery Manages the Energy Flow

The anode releases electrons to the negative terminal via the circuit, and ions through the electrode, as it discharges. The cathode accepts these simultaneously, thereby generating an electric current. When the anode runs low on ions, the battery is flat. If it is a single-use disposable one, then that is the end of its useful life.
We can replenish a rechargeable battery by connecting it to an external power source. This sends the ions back to the anode, restoring its electrical potential.
However, each time we discharge and recharge a battery, some ions become stuck in the electrolyte. This is how a battery deteriorates, and reaches the end of its life. That’s all there is in principle to how batteries work.
Related
How Electrons Travel In and Out of Electrodes
Preview Image: Disassembled Zinc Chloride Cell