We use the term ‘electro-chemical’ because this article is about conventional batteries. Our scope therefore excludes gravity, water, and hydrogen storage, as well as other exciting innovations. Let’s get down to explaining exactly how do electro-chemical batteries work right away. Because these tiny cells have an essential role to play in mitigating climate change.
Electro-Chemical Cells Are Chemical Reactions
A conventional battery – including alkaline, nickel, lithium-ion and lead-acid chemistries – converts chemical energy into electrical energy. A battery can contain one or more electro-chemical cells, but the principals remain the same.
We need to understand what electricity is, to get a grip on how electro-chemical batteries do their work. To put this simply – so science does not hold us up – electricity is energy created by a flow of electrons.
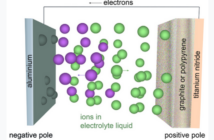
This flow of electrons originates in a chemical reaction occurring at one of a battery’s two electrodes. These electrons flow to the battery’s second electrode via an external circuit, where some of them are consumed.
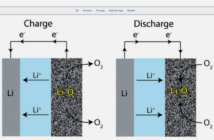
When all the battery’s electrons are used up, the battery is ‘flat’. However, if the battery is a rechargeable one, then we can add a fresh supply of electrons from a battery-charger drawing electrical energy from an external source.
The Electrolyte Balances the Charges in a Battery
A bolt of lightening striking earth confirms that for every reaction there’s a balancing counter-reaction. The electrons leaving a battery from the first to the second electrode have a negative charge. This disturbs the polarity of both electrodes.
To compensate for this, the electrolyte separating the electrodes generates a flow of positive ions. These balance the polarity in the battery so the reaction can continue. However, if we disconnect the battery from the external surface, then the chemical reaction almost entirely stops.
More Information
Battery Operating Basics in More Detail