This post continues on from an earlier one (see first link below), where we considered the components of electro-chemical batteries. You may recall these constituents include a pair of electrodes, plus an electrolyte through which their ions pass. We drill down into battery operating basics in this second post, in order to compete a broad view of our product.
Fundamental Operating Principles of Batteries
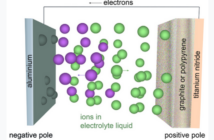
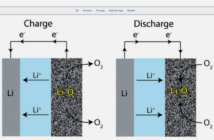
The negative, or reducing anode battery terminal is usually a metal, able to oxidize and surrender electrons. While the positively charged cathode is able to accept these electrons, and is most likely to be a metal oxide or a sulfide.
However, these battery operating basics could not exist without an external circuit able to transport electrons between the terminals. But even so, this process could still not complete, without an electrolyte containing chemicals inside the battery.
That’s because this electrolyte provides the pathway which ions travel between the electrodes. And thereby offsets the flow of current outside the battery, through the external circuit, thereby balancing the charge
Factors Affecting Battery Operating Performance
Not all batteries perform at the same level. Indeed, their chemical constituents are carefully selected to achieve particular goals. The chemicals in the electrolyte, for example, must contain salt ions able to move sufficiently fast to achieve a required electrical standard.
The ambient temperature around a battery also has a bearing on its performance. Ideally, this should be at a level humans find comfortable. However, if the temperature chills considerably during a polar vortex, for example, then the battery may cool and produce far less current.
If the temperature increases, on the other hand, then the battery may return to its original state unless damage occurred. But if that temperature becomes excessive, then the battery may suffer irreversible damage. Your take away from this post could be to operate your battery within a range of temperatures where you are comfortable too.
More Information
Getting Back to Battery Basics for While