Redox is the abbreviated short form of ‘reduction-oxidation’, which occurs as electrons transfer between substances at molecular level. The article will assist you if you need to know how redox works in batteries in simple terms.
When a battery discharges:
- The anode electrode loses electrons during the ’oxidation’ process.
- These electrons travel through an external circuit to the cathode.
- The cathode gains those electrons during the ‘reduction’ process.
Let’s Dive a Little Deeper Into Battery Redox
At this stage you may be wondering how anything can gain something through reduction. The straightforward answer is ‘reduction’ comes from the Latin word ‘reducere’, which means to revert to an earlier state.
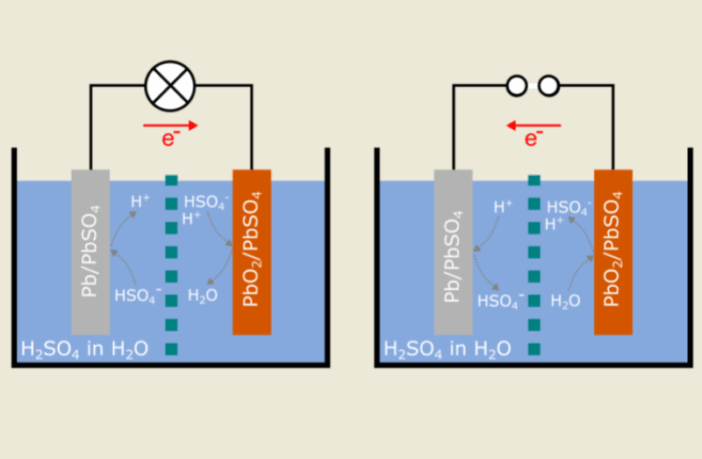
Getting back to the basics of how redox works in batteries, let’s examine the process in lead-acid batteries as they discharge:
- The spongy lead (Pb) at the negative plates OXIDIZES to become lead sulfate (PbSO₄), as it gives off electrons.
- The lead dioxide positive (PbO₂) plates receive these electrons, and REDUCE to become lead sulfate (PbSO₄).
- These processes weaken the sulfuric-acid / water electrolyte, meaning the solution becomes less efficient.
To recap then, we end up with sulfate at the cathode and anode, and less sulfuric acid in the electrolyte. In return we get electricity from the battery. When we recharge the battery we put the electricity back again.
Recharging a lead-acid battery reverses the discharging process, as it completes the second half of the reduction-oxidation cycle. This is what happens when we apply electricity from an external source:
- We send the electrons back to the negative anodes. This REDUCES them to their near-original state.
- In parallel, the lead sulfate at the positive plates OXIDIZES to become lead-dioxide again.
- Both plates surrender their lead sulfate in the process. They return it to the electrolyte, strengthening the solution to its near original state.
This completes our introduction to how redox works in batteries, using lead-acid chemistry as an example. Did this help you understand the redox concept better?
More Information
Lead Batteries Indispensable in EVs