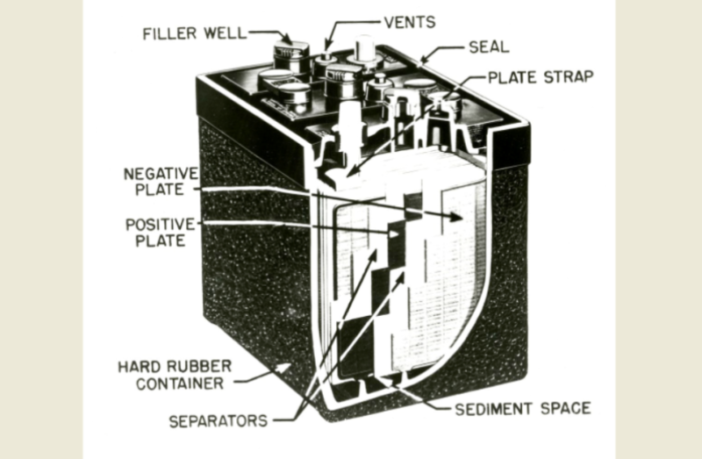
Lead-acid batteries – almost all batteries in fact – comprise an anode, a cathode, a separator, and electrolyte. Separators feature far less in the media than the other three components. So today we ask what role does a lead-acid battery separator play, and how did they evolve. You may like to read on, and discover details you may not have known.
Brief Introduction to Lead-Acid Battery Separators
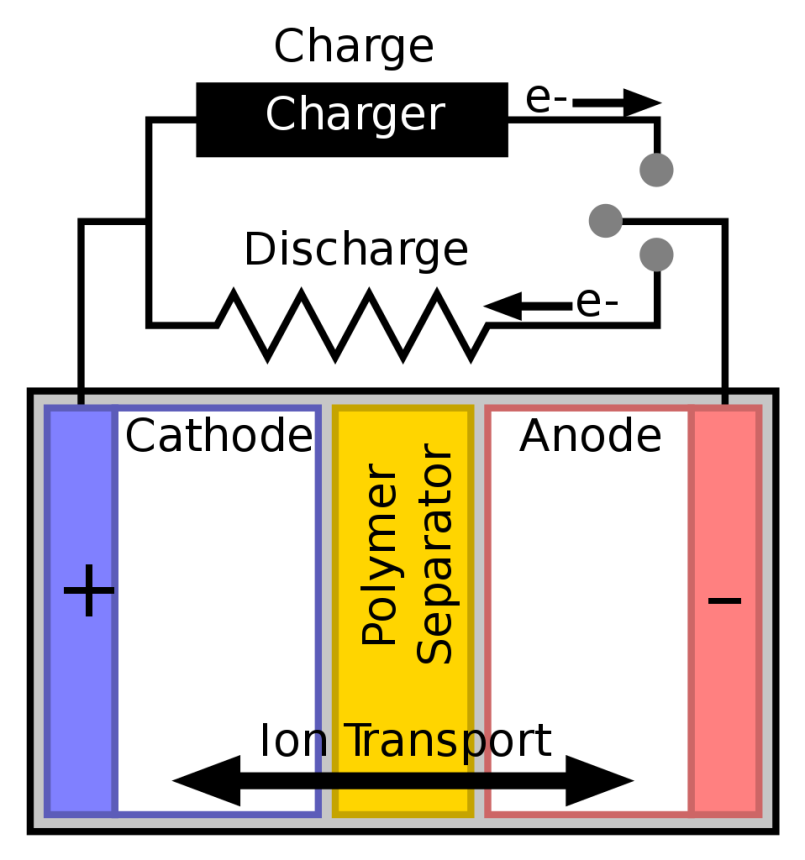
Battery separators are polymer derivatives inserted between positive anodes and negative cathodes. This prevents those two electrodes from touching, and helps prevent them electrically short-circuiting.
But if the electrolyte moistens a lead-acid battery separator, then it also facilitates the movements of ions from cathode to anode as the battery discharges.
A Short History of Battery Separators
French physicist Gaston Planté invented the first rechargeable battery in 1859, and it was a lead-acid one! That version used a wet cell / flooded design, without a separator according to Hollingsworth and Vose. In fact, the lead-acid battery separator only arrived with the introduction of maintenance-free lead-acid batteries in the 1970s.
These more advanced lead batteries featured electrolyte-saturated, porous polymer separators. They pressed firmly up against the electrodes, kick-starting the chemical reaction. The first prototype separators were made from wood. But today sealed lead acid batteries rely on glass fiber mats soaked in sulfuric acid.
Quality Criteria For All Battery Separators
Wikipedia defines the following standards regardless of the battery chemistry type:
- Chemical stability with less likelihood of degrading.
- Optimum thickness to facilitate many charging cycles.
- Sufficient porosity to hold adequate liquid electrolyte.
- Pore size smaller than electrode component particles.
- Adequate permeability to allow electrons to flow through.
- Mechanically strong enough to fulfill its separator tasks.
Battery separators are critical components in liquid electrolyte batteries. Their structure and properties markedly affect battery energy and power density, as well as cycle life and safety.
More Information
Battery Separator from Seaweed Nanoparticles