We know that batteries charge and discharge, by moving charge-carrying ions between their electrodes. Today we ask two fundamental questions. What exactly are these charge-carrying objects, and what is the origin of ions in batteries. These are far-reaching issues, for without ions we could not have electrochemical batteries.
The Origin of Energy in Batteries
An ion is a single atom, or a small group of atoms, carrying an electrical charge. Most atoms are electrically neutral though, because they contain equal numbers of positive particles (protons) and negative particles (electrons).
This stable situation changes when an atom loses or gains one or more electrons, to or from another nearby atom. This means it holds a net negative or positive charge, and therefore becomes an ion:
- If an atom loses one or more electrons, the net charge is negative and we call it an anion.
- If an atom gains one or more electrons, the net charge is positive and we call it a cation.
Those charged ions could be lithium or sodium atoms, or small groups of atoms, just as an example. They matter a great deal in battery chemistry, because ions can move through certain materials, including special liquids and solids.
The Role of Ions In Batteries
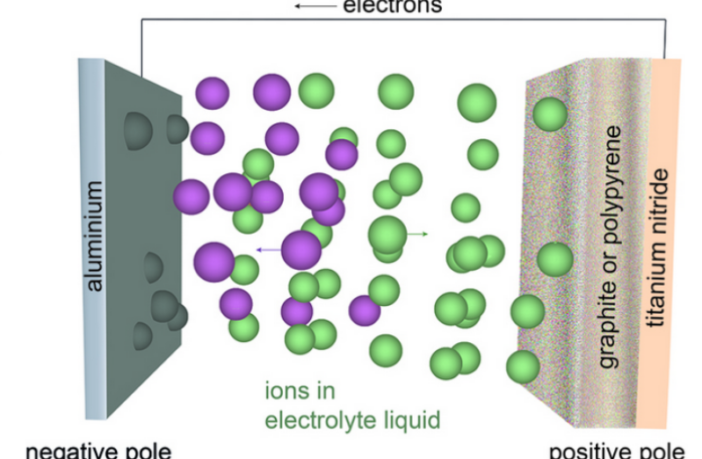
We now understand the origin of ions in batteries, so lets turn to what they do. Each electrochemical battery comprises a pair of electrodes, and a liquid or solid electrolyte material between them. Now ions, as we know, can move through certain liquid and solid materials.
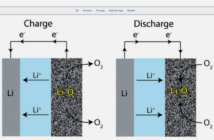
When a battery comprising compatible materials operates, a chemical reaction begins at one electrode. This reaction produces ions that travel through the electrolyte to the other electrode. At the same time, electrons move through an external circuit such as a phone, in the opposite direction.
This movement of ions inside a battery keeps the chemical reactions balanced, while electrons flow outside the battery to produce useful electrical energy. Without this internal ion movement, the battery could not keep supplying electricity that way.
When a battery runs down, the chemical materials that produce the ions are mostly used up. In rechargeable batteries though, applying an external electrical current returns the ions in the opposite direction. This should restore the battery to its near-original state, all things being equal.
More Information
The Movement of Cations and Anions