The very first battery was a byproduct of an experiment, an accidental event that almost passed by unnoticed. There are many other similar examples in the annals of science, but this does not diminish their value. After all, how can we deliberately look for something if we do not already know that it is there? This is what makes science so exciting, and sometimes so dramatic too.
The Very First Battery Was an Accident
Luigi Galvani was an Italian physician, physicist, biologist and philosopher, with a particular interest in animal electricity. We find this energy in all living beings. In fact, it drives basic processes such as muscle contractions, and nerve conduction in humans too.
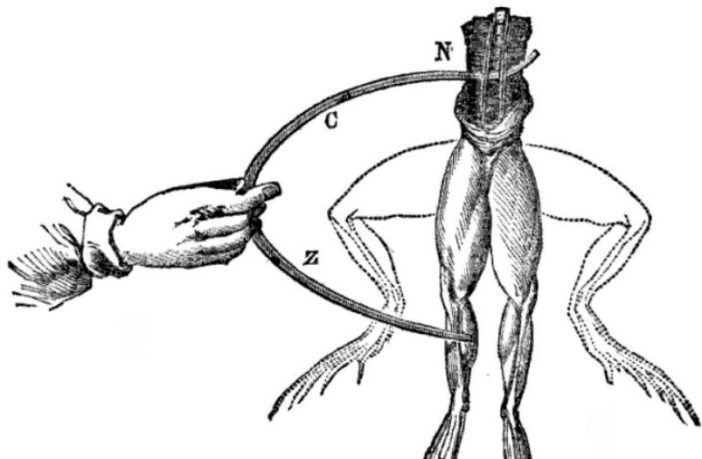
One day in the late 1780’s Luigi Galvani conducted an experiment, during which he accidentally created the world’s first battery we know of. He did so by hanging a dead frog’s legs on a brass hook. However, this was only half the battery …
Luigi ‘completed the circuit’ by touching one leg with a probe that was of a different metal from the hook. He believed he had demonstrated animal electricity when the dead frog’s leg twitched. However, his colleague Alessandro Volta saw things differently!
That was because Alessandro was a physicist and chemist interested in electricity and power, and therefore saw things another way. He realized Luigi had actually transmitted electric current between the hook and the probe. In modern battery terms, the frog was merely the electrolyte!
Applying These Principles to Science
Volta realized the clue lay in the fact the hook and probe were of different metals. He replicated the phenomenon with a pile of alternating silver and zinc discs (the electrodes) with layers of cloth or paper soaked in saltwater between (the electrolyte). Electricity flowed through his battery as he predicted, after he applied a wire to both ends.

Breaking News
Electric Vehicles Are Older Than We Think