Life would be chaotic if ions could travel in any way they wanted inside batteries. Electrolytes control this situation, by shepherding ions between battery cathodes and anodes when we recharge them. And then managing the reverse flow when we connect the battery to a device, and discharge it. We review common types of battery electrolytes, because different chemistries require different solutions.
Battery Electrolytes – The Commonest Types
There are several generic types of electrolytes, which engineers tweak to suit particular applications. Broadly speaking:
- Electrolytes comprise soluble salts, acids, or other bases.
- These alternatives may be in liquid gel, or dry formats.
- But they may also be polymers, solid ceramics, or molten salts.
LEAD-ACID BATTERIES
Lead-acid batteries use highly corrosive diluted sulfuric acid as their electrolyte. This pure acid has a slight yellow-green tint, and is soluble in water. However, the diluted version may develop a brownish tint, from corrosion at the anode.
When we charge a lead-acid battery, lead oxide forms on the positive plate, causing the electrolyte to become denser. The process reverses during recharging, causing the battery to reinstate. However, some oxidation may remain, gradually weakening the battery.
NICKEL-CADMIUM AND NICKEL-METAL ELECTROLYTES
Nickel-cadmium and nickel-metal batteries use alkaline electrolytes, which are usually compounds of potassium hydroxide. This common type of battery electrolyte is an inorganic compound, commonly referred to as caustic potash. The material is generally harmless as long as we do not ingest it, and it is an ingredient in most soft and liquid soaps.
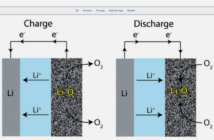
ELECTROLYTES IN LITHIUM-ION BATTERIES
Lithium-ion batteries use liquid, gel, or dry polymer electrolytes. The liquid version is flammable, being a blend of lithium salts and organic solvents. There are many variations to this theme, and research is ongoing as we publish it here.
Lithium-ion batteries are generally stable, although they may overheat if they exceed their operating specification. For this reason they should never be left on their own when recharging, or abused or damaged in any way.
More Information
The Ideal Size of a Home UPS Battery