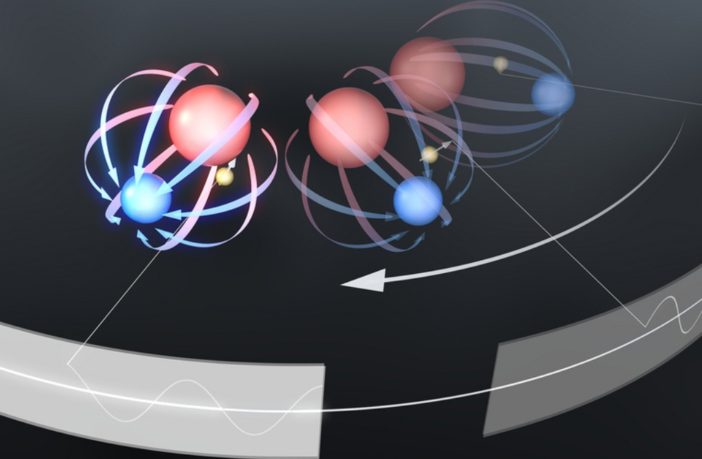
Chemical reactions in batteries, fuel cells, and human cells have something in common. That’s because they all feature association and disassociation of ion pairs in water. Researchers at the Physical Science Division of Pacific Northwest National Laboratory have recently derived an elegant method to describe how water helps separate ions in this way.
How Water Helps Separate Ions and Draw Them Together
Ion pairs in water affect chemical reactions inside fuel cells, batteries, and biological systems. This research shows how water moves around ions and causes them to draw together or stay apart, and provides insight into how they pair.
Traditional computational models have to date failed to discern the underlying mechanism. The scientists at Pacific Northwest National Laboratory have come up with a theory to explain this. They believe “water moves around pairs of ions and influences whether they draw together or stay apart,” according to Phys.Org.
Disassociation of Ions Takes Place in Two Stages
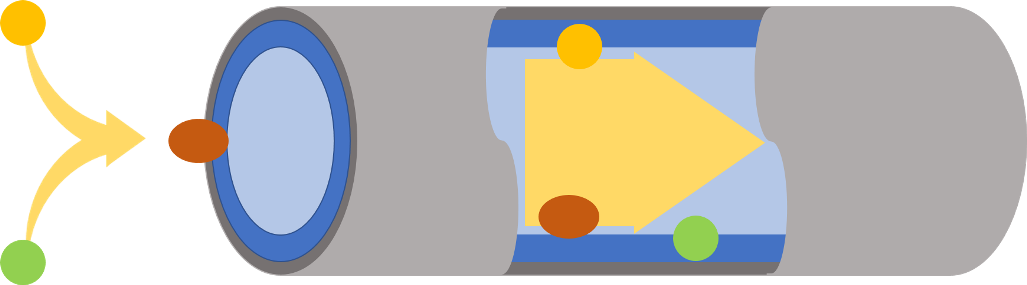
The scientists began their research by observing the distance between ions. And the number of water molecules around either an individual ion or an ion pair. From this, they deduced that ion disassociation occurs in two stages.

First, there is an increase in the number of water molecules around each ion. Then secondly, the ions move apart as the water molecules move away. Water moving is the critical, rate-limiting step, they say. This framework builds on a theory originally developed by Rudolph A. Marcus in 1956.
His theory explains the rate at which an electron can move or jump from one electron donor to another called the electron acceptor. This new contribution offers “an improved understanding of ion pairs. From protein interactions with DNA, to ion motion in batteries” Phys.Org advises.
Related
MXenes Could Grow Charge Life By 40%
Secret Life of Lithium-Rich Batteries Revealed
Preview Image: Electrons in Motion