The battery inside your phone has a positive and negative ‘terminal’ on the outside of the case. The terminals connect to ‘electrodes’ inside, which contain the electricity that powers your device. Today we explore what is in the cellphone battery itself, and share a few tips to keep it working safely, longer.
What About the Chemistry Inside the Cellphone Battery?
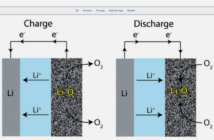
Good point! Scientists call batteries ‘electrochemical cells’, because they use chemistry to produce electricity. This energy (which is what electricity is) resides in atomic particles called ‘ions’ with a net electric charge. These ions travel between the electrodes as the battery charges and recharges.
But that’s not all there is to say on the topic of what is inside the cellphone battery you use each day. That battery also contains a control mechanism we call an ‘electrolyte’. This material (which may be liquid or solid) insulates the terminals from each other, but allows the ions to travel between them.
We can recharge a smartphone battery repeatedly many times, provided we do this correctly. Or perhaps should we say return the spent ions to the storage electrode, so we can use them again. The electronics in phones are sensitive. Always use the charger that came with the phone to ensure the current is spot-on.
Lithium Phone Batteries Require Extra Precautions
Most smartphone batteries contain a silvery metal called lithium. This is the third lightest element after hydrogen and helium, and is inherently unstable. Lithium responds to high heat by catching fire, and this is not easy to extinguish:
- Use the right phone charger to avoid overheating the battery.
- Replace a cellphone battery immediately you notice damage.
- Treat your phone with care, so you don’t accidentally damage it.
TIP: Cellular data uses more battery power than Wi-Fi, because of the energy consumed searching for a cellular network signal.
More Information
What to Do When Smartphone Batteries Swell