Lithium-ion batteries have become a part of our day to day life. We use them in our laptops, mobile phones, toys for kids, many electronic gadgets and even in our electric cars. This is because we can charge them repeatedly.
The lithium-ion batteries conventionally use Graphite based anodes. These graphite based batteries are slow in charging and discharging. They take hours to get charged completely. Also, they are quite bulky. They constitute a significant portion of volume and mass in electric vehicles.
To overcome the shortcomings of graphite based batteries, researchers at University of California have come up with a solution. They have developed a Three-Dimensional, Silicon based, Cone shaped, Carbon Nanotube Cluster Architecture for Lithium-ion batteries. This silicon based architecture promises to charge batteries in 10 minutes flat.
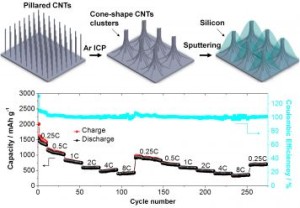
Image Courtesy: http://ucrtoday.ucr.edu/
Features of new silicon based architecture
Silicon is a type of anode material that has 10-times higher charging capacity than commercial graphite-based anodes. If the graphite anode is replaced by a silicon anode in a packaged battery, the total cell capacity can increase by 63%. If we retain the old capacity, the battery size would be reduced by 40%.
Breakthrough Technology
The new architecture exhibits highly reversible capacity along with brilliant cycling stability. Such batteries show exceptional electrochemical stability even at high charge and discharge rates. These rates are 16 times faster than the conventional graphite based model.
According to the scientists, this very high rate of charge and discharge of the battery is due to two factors. First, there is a seamless connection between graphene covered copper foil and carbon nanotubes. This seamless connection boosts active material-current collector contact integrity. This means that graphene covered copper foil acts as a catalyst by reducing the gap and increasing the level of contact between active material that is lithium and carbon nanotube which acts as the current collector electrode. This leads to better charge and thermal transfer in the electrode system.
Second, the Cone shaped architecture model provides small interpenetrating channels. These channels lead to faster electrolyte access into the electrodes, thereby enhancing performance.
The world is waiting with bated breath for this technology to be applied commercially in all Lithium-Ion batteries.
Related Posts
What are Lithium-Ion Batteries
Carbon Nanotubes: Replacing Silicon in Circuits


