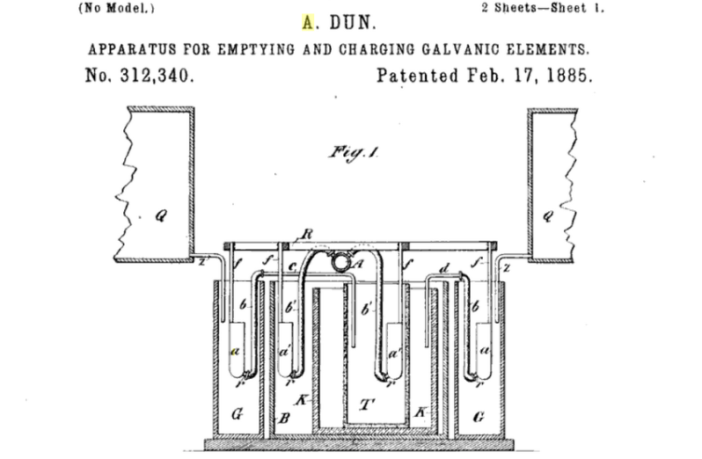
Alfred Dun is a shadowy figure, and something of a mystery in the annals of battery history. He appears in an 1885 U.S. patent specification, where he describes himself as residing at Frankfort-in-the-Main in Germany. He explains his Dun Cell is primarily for electric lighting, but it has other uses too. That’s about all we know about him. We shall do our best to unravel how his battery cell worked.
The Operating Principles of the Dun Cell
Alfred Dun described his invention as a galvanic element in his patent application. In other words a type of simple electric battery. It comprised two electrodes, and a nitro-muriatic electrolyte he called the ‘exciting fluid’.
The positive electrode was iron, while the negative one was carbon. However, what makes this design stand out was the composition of the electrolyte solutions.
- The positive cathode electrode used a highly-diluted nitro-muriatic solution.
- While the negative anode electrode had almost pure nitro-muriatic electrolyte.
Alfred Dun explained this particular ratio delivered a ‘very constant battery’ of a caliber similar to using two different electrolytes. In fact, he claimed the output ‘remains constant for at least twenty hours when employed for electric incandescent lighting’.
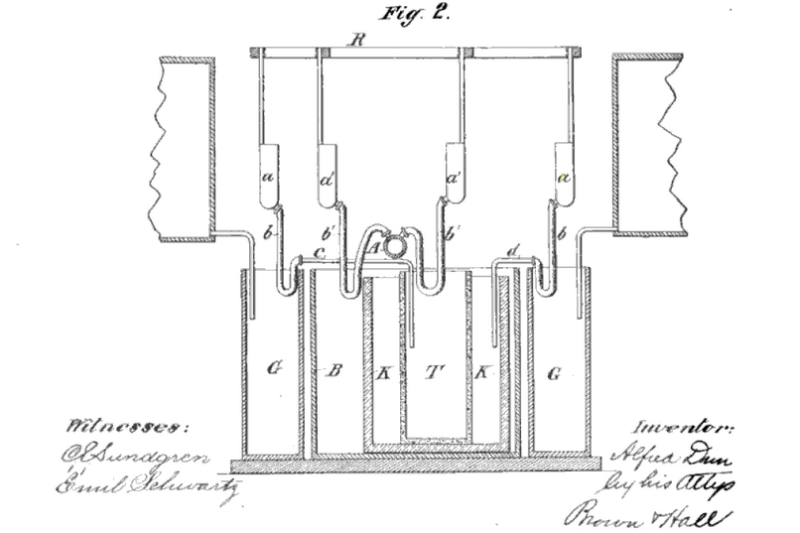
The Battery Therefore Had Two Separate Cells
We can glean from the patent specification (see link below) that the Dun Cell was bi-cellular. This puts it in the same ball park as the Daniell Cell and its derivatives. But Alfred Dun claimed his design was an improvement over them, because there was no possibility of cross-contamination between electrolytes. And moreover the stronger electrolyte could dilute as the weaker electrolyte, when it was wearing down,
We will probably never know how successful Alfred Dun’s invention was, or indeed whether he prospered from it either. That’s because about the closest we got when googling Alfred Dun, was articles about a tobacco company. We owe much to the silent scientists laboring for their own work’s sake. They may never make to the halls of fame, but they do deserve their places in history.
More Information
Experiments in Science: The Grove Cell
A Porous Pot Cell in a Copper Can