Did you ever wonder where the ions go after they shuttle between the electrodes, and release their electrical energy? One of the facts of intercalation is they slip between the layers of the material making up the anode. And then return to the anode when it is time for them to recharge, like fingers slipping back into a glove.
Intercalation is a Basic Fact of Electro-Chemistry
This simple fact of intercalation is the key to all portable electronics, for without it they would not be possible. Neither would the batteries for that matter that power their circuits. All of battery energy and power density, safety, and stability depend on the composition of the anode and cathode electrodes, except in the case of lead-acid batteries.
However, battery efficiency is still a bottleneck to achieving universal, grid-scale renewable energy. When you were a kid and you held a flashlight battery in your hand, we’ll bet you had no idea how important these things would become, indeed for the future of our planet.
This simple fact of battery life we call intercalation is the key to every electric automobile, and part of the reason behind our ability to store renewable energy. Without it, there could be no smart watches, smart phones, and life-preserving pacemakers. Stanley Whittingham discovered the mechanism in the 1970’s, and this enabled him to develop the original lithium-ion battery.
That initial push was driven by anxiety over warming, a concern so few people shared. Nowadays, of course, it is global knowledge and the call is for more batteries with greater capacity storage.
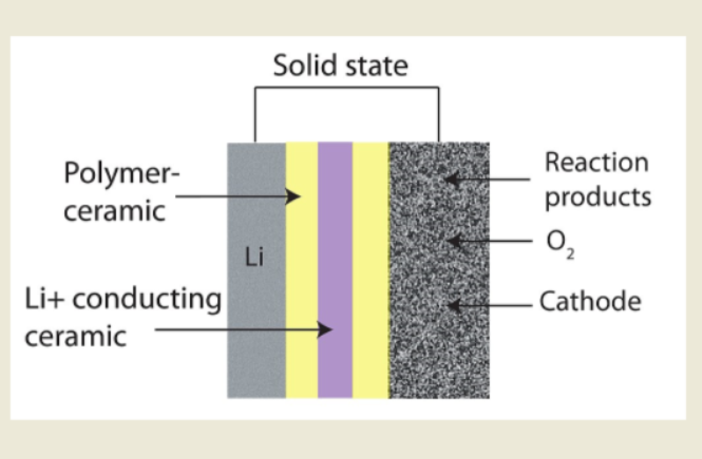
Whittingham’s batteries are still making a tremendous contribution, although they do have their drawbacks. However, despite all the money and all the research, few alternative practical cathodes and anodes exist. The next step forward could be lithium-air batteries. These do away with intercalation, as we know it, by exchanging ions with the air.
More Information
Smart Electric Water Heating Technology