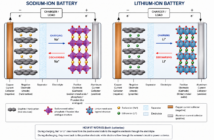
The solid electrolyte interface (SEI) is a passive layer that forms between the electrolyte and the graphite anode in lithium batteries. The SEI is the magic sauce that makes lithium-metal batteries rechargeable. Scientists at National Renewable Energy Laboratory researched how this interface layer eventually dooms the chemistry to fail.
Photographing Solid Electrolyte Interface Secrets
Flat coin cell battery materials are so tightly squeezed that it is difficult to observe what is going on inside. Scientists at National Renewable Energy Laboratory froze a lithium-metal button battery. Then they opened the battery up with a super-fast laser, and photographed the interacting components.
Their microscopic images revealed how lithium-metal batteries fail sooner than lithium-ion ones, during charging and discharging. This negative characteristic has side-lined a chemistry that stores more energy than lithium-ion does. Many scientists believe the key lies in the solid electrolyte interface.
The Untapped Potential of Lithium-Metal Batteries
Using lithium-metal chemistry could lead to lighter, smaller batteries. First though, we would need to find ways to counter their limited cycling and consequential safety issues. One persistent theory holds that the lithium spikes through the separator. Then its energy morphs into heat and starts a fire.
But the National Renewable Energy Laboratory saw something different when they froze a battery, opened it, and observed events under high magnification:
- The lithium reacted with the liquid electrolyte to form solid masses.
- These solid masses shredded the battery separator ‘from inside out’.
This information will help guide further battery research, scientists believe. Moreover, it should help reveal ‘more mysteries that occur in other complex, solid-liquid environments’.
What This Taught Us About the Solid Electrolyte Interface
This research enabled scientists to observe lithium-metal battery components for the first time, without damaging them. Thanks to this work we now know the solid electrolyte interface either obstructs lithium movement, or creates a trail that leads to catastrophic failure.
More Information
Resting a Battery Restores Lithium-Metal
Solid–Electrolyte Interphase – Dramatic News