Scientists at University of Michigan have invented a stabilizing coating on lithium-ion battery electrodes, that makes a remarkable improvement. Their innovation allows rapid electric vehicle charging, at temperatures below freezing. Charging EVs 5x faster in subfreezing weather, could resolve the trade-off between charging speed and freezing temperature, when we would rather be tucked up in bed.
Adapting Manufacture to Allow Ultra-Rapid EV Charging
Associate professor of mechanical engineering and materials, science and engineering Neil Gasupta, has good news for electric vehicle battery makers. “We envision this approach as something that EV battery manufacturers could adopt” he explains, “without major changes to existing factories.”
This appears to be the first time there is a pathway to extremely-fast-charging lithium-ion batteries at low temperatures. While at the same not sacrificing any of their all-important energy storage density, which is the reason we may use them.
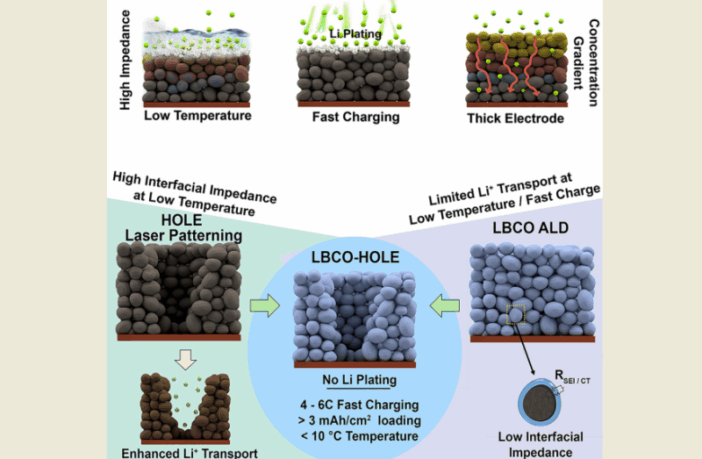
We understand that manufacturing lithium-ion batteries in this way, will facilitate charging EVs 5x faster, in subfreezing weather even as low as 14 F (-10 C). The structure and coating the team applied to their lithium-ion battery electrodes, prevented lithium plating which normally degrades performance.
This modification allowed the prototype battery to discharge and recharge 100 times, while retaining 97% of its original capacity, even at very cold temperatures. But how did a stabilizing electrode coating achieve this?
How Did the Coating Facilitate Fast Subfreezing Charging?
Lithium-ion EV batteries store and release their power, through the movement of lithium ions between electrodes through a liquid electrolyte. The movement of these ions slows down in cold temperatures, reducing battery power and charging speed.
Increasing electrode thickens helps somewhat, but it also makes the battery heavier. The University of Michigan team decided that the lithium plating was actually the root cause of the problem.
They had previously tried drilling holes through electrodes with lasers, but this only improved things somewhat. And so they coated those same modified battery electrodes with a glassy material made of lithium borate-carbonate, approximately 20 nanometers thin.
Combining the two methods allowed the prototype battery to charge 5 times faster in subzero temperatures. This sounds so simple, but then again, many things are in hindsight, are they not?
More Information
Rapidly-Charging Freezing Lithium Batteries
Michigan UNI Doubles Up Battery Research
Preview Image: Improving Subfreezing Charging