Lithium has pretty much dominated battery space this past decade. We have much progress to thank it for. But lithium does have its challenges. In the first place, overheating is possible because lithium is unstable and flammable.
In addition there is also the question of cost. For these reasons aluminum-graphite batteries are knocking at lithium’s door.
Aluminum-Graphite Batteries’ Fundamentals
An aluminum-graphite battery has an aluminum anode and graphite cathode. This provides an impressive pedigree of power potential over lithium.
- 50% cheaper in terms of production
- Specific density is 1.3 to 2.0 times higher
- Energy density is 1.6 to 2.8 times greater
Incredible Charging Potential of Aluminum-Graphite Batteries
But that is not all. Researchers at Stanford University in California have built an aluminum-graphite smartphone battery. This has capacity to recharge in sixty seconds. Sure, it only holds its power half as long as lithium. But with such a fast recharge time, this doesn’t sound a problem.
Move over, free wi-fi and laptop stations in fast food restaurants. Imagine recharging outlets for aluminum-graphite batteries everywhere we go. Visualize dropping a quarter dollar into a slot on a subway train and having your battery back in one minute.

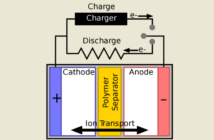
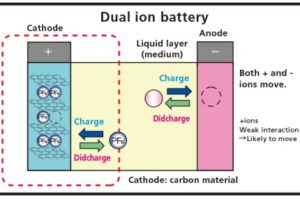
Technology of Aluminum-Graphite Dual-Ion Batteries
Dual-ion batteries have low internal resistance. This permits fast charging and discharging. This is practical even where high current regenerates from large machines like electric cars. The Ricoh graphic illustrates this principle. At this stage it is still using a carbon cathode.
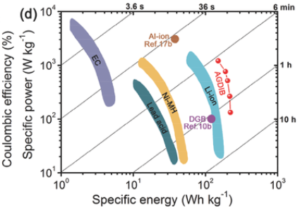
Here is a scientific report on work on experimental aluminum-graphite batteries in dual-ion format. Chinese scientists are using ethyl–methyl carbonate electrolyte. This works around a tricky challenge.
Firstly, finding ‘a suitable electrolyte enabling both Li+ intercalation into a graphite negative electrode. Then having intercalation into a graphite positive electrode simultaneously.’ This sounds like having two batteries in one for half the cost, and a fraction of the charging time!
Related