We use the word ‘currently’ not in the electrical sense, but in the context of the present. This because lithium-ion technology is developing at such a rate we dare not predict the safety winner. Thus we are experimenting with inherently unstable isotopes. It is a bit like riding a tiger.
Conventional Lithium-Ion Technology in Practice
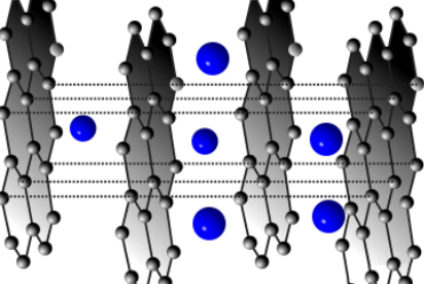
Lithium-ion technology manages the inherent instability of reactive lithium metal. It avoids its presence during the charge-discharge cycle. Instead, it moves lithium ions between the electrodes. We call the process intercalation, or the reversible insertion of a molecule into a compound.

The lithium ions are in the positive electrode when a lithium-ion battery is in fully discharged state. Conversely, they are in the negative electrode when the battery holds a full charge.
In practice, some lithium-ion molecules are present in both electrodes. Scientists compare their constant shuttling to a rocking chair moving to and fro.
The positive anode contains lithium ions intercalated into carbon and occasionally metallic lithium. The negative cathode is a lithium-liberating compound of cobalt oxide, manganese oxide, or nickel oxide. Gel or liquid electrolyte in a thin film separates them. If that fails, the lithium overheats. This is the ‘achilles heel’ of lithium-ion technology.
How Lithium-Ion Polymer Technology is Simply Safer

Lithium-ion polymer batteries are inherently safer. In fact, they represent the currently most stable lithium-ion technology. Their electrolyte is a solid polymer material that theoretically cannot leak. For this reason, they carry the generic name ‘solid state cells’.
Polymer lithium-ion technology produces long storage life batteries with low discharge rates. They are lighter than conventional lithium ion because there is no need for a protective case. With them, we can create shapes that suit our application. This makes them ideal for mobile phones, wearables, and ‘toys-for-boys’ including radio control aircraft and trains.
Related