Intercalation describes a process whereby molecules, or ions, insert into layered materials with layered structures. We could compare intercalation in batteries to slipping playing cards between the pages of a book, but there the similarity ends. Not all materials have these layered structures suitable for batteries.
Intercalating Charged Battery Ions in Graphite
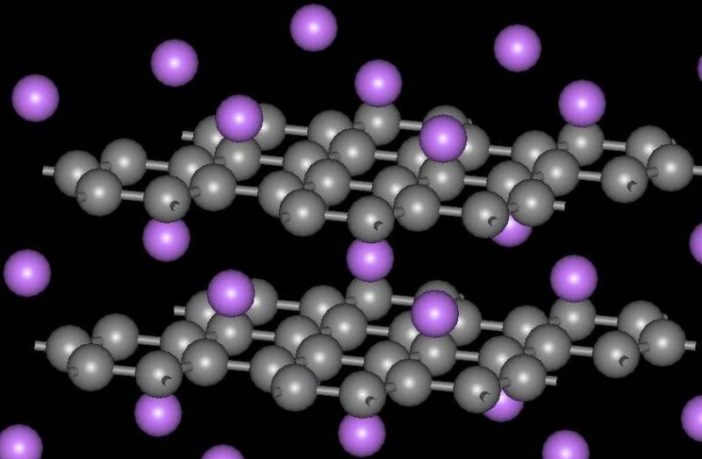
Graphite is the most popular layered material in our world of batteries. Intercalating ions use their energy to stretch the gap between the layers. This power transfers from the charged ions to the layered electrode during intercalation.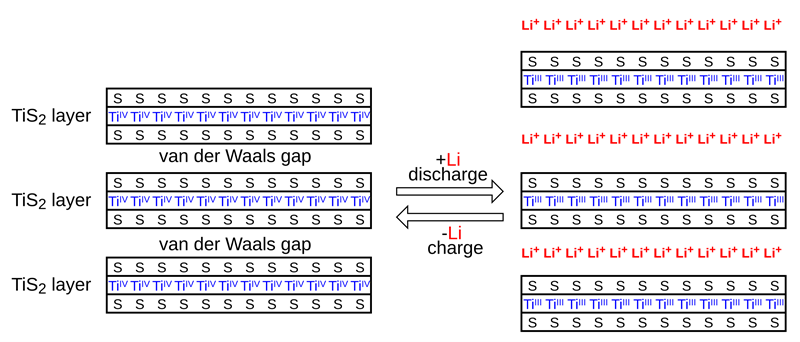
Intercalation of Lithium Into a Titanium Disulfide Cathode (Д.Ильин BY CC 4.0 Share Alike)
Intercalating molecules between layers in materials increases their volume, and stresses their crystal structure. The prevailing temperature, and the rate at which this happens can increase these stress levels.
Side Effects of Intercalation in Batteries
Intercalation is the power behind emerging battery technology, notably lithium-ion and sodium-ion chemistry. However, this process causes a gradual accumulation of residual stress. This may cause mechanical fatigue, and microscopic cracks at stress points.
This mechanical degradation progressively diminishes the amount of energy in a battery, as a result of several adverse electrochemical events:
- Electrical conductivity reduces within the active material in the battery.
- Active molecules are lost due to the formation of the SEI interphase.
- There is increased potential for cracking, and formation of dendrites.
Intercalation in batteries thus gradually destroys the power in the very batteries that it enables. Much research effort is ongoing to retard this process.
Implications for Battery Design and Research
Battery designers reduce intercalation stress, by choosing electrode materials compatible with intercalating ions. This is why lithium cobalt oxide, and lithium iron phosphate are popular in lithium batteries.
Solid state battery technology may have eliminated some of the issues associated with liquid electrolytes. Notwithstanding this, solid electrolytes may also accumulate mechanical stress, as molecular ions pass through them.

Volumetric expansion in solid state batteries can also result in poor electrolyte-electrode interfaces. Stress resulting from intercalation in batteries remains a persistent problem, affecting battery performance and battery life. Visit us often for more updates!
More Information
Co-Intercalation for Faster Sodium Charging
Understanding Intercalation In Batteries 101