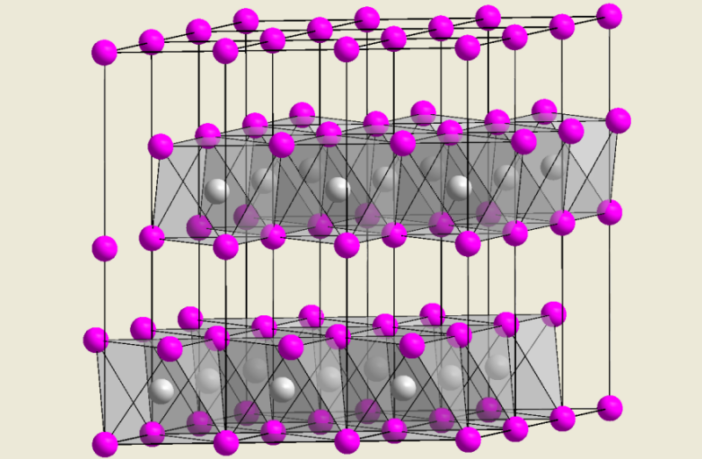
Graphite and some other materials comprise two-dimensional sheets, that are only weakly bonded to adjacent ones. This weak bonding allows the reversible insertion of molecules or ions into their spaces. Understanding intercalation in batteries is critical, if you really want to know what happens inside your phone.
Intercalation Completes Our Understanding of Batteries
The process that follows occurs under the influence of Van der Waals principles. These govern the attraction of intermolecular forces between molecules.
An energy source is required to expand graphite’s weakly-bonded spaces. This energy usually arises from redox charge transfers between guest and host solids. Such redox reactions are part of a battery’s normal charge and discharge cycle.
John Goodenough applied these principles to batteries in the early 1980’s, and deserves much credit for the modern lithium-ion battery. He, his predecessor Stanley Whittingham, and Akira Yoshino who followed after him, jointly shared the 2019 Nobel Prize in Chemistry, for their research into lithium-ion batteries.
Intercalation Theory and Practice in Lithium-Ion Batteries
John Goodenough applied his new understanding of intercalation to developing a prototype rechargeable lithium-ion battery. Since then, these energy storage devices have become commonplace in portable electronic devices, electric vehicles, and utility-scale renewable energy storage.
By 2023, all commercial rechargeable lithium-ion battery cells used active intercalation compounds, most of which were in both the anode and cathode electrodes.
Lithium-ion has been a great success story, and is fundamental to rolling out green energy. However, the chemistry is not ideal in terms of negative social and environmental consequences. In five year’s time, a new battery chemistry may have taken its place. We shall have to wait, watch, and see what happens.
More Information
John Bannister Goodenough Battery Pioneer