Sodium–zinc molten salt batteries are a relatively new type of high-temperature energy storage system. Their main strength lies in delivering reliable renewable electricity to utility distribution grids.
They Are Part of a Team of Molten Salt Batteries
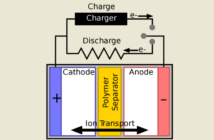
The sodium-zinc version uses sodium in the negative electrode, and zinc in the positive one. The term ‘molten salt’ acknowledges the fact that these two metals are in a heated, melted state.
To complete the picture, the complementary electrolyte typically becomes conductive at 250 °C to 350 °C / 480 °F to 660 °F.
When sodium–zinc molten salt batteries reach these temperatures, their zinc and sodium metals become reactive. This means their ions can move efficiently through the electrolyte, as they store and release energy.
More About Sodium–Zinc Batteries With Melted Salts
The two electrodes have a solid ceramic electrolyte between them, that prevents them touching but allows them to exchange ions.
- Their sodium atoms release electrons and form sodium ions, while they discharge. The ions travel through the electrolyte to react with zinc at the cathode.
- This process reverses when the battery recharges, so the system can store electrical energy at the anode. This completes the electrochemical cycle.
Factors That Make These Batteries Interesting
Sodium–zinc molten salt batteries have the advantage of using abundant and relatively low-cost raw materials. These are widely available, and environmentally less contentious then lithium-based technologies.
This means our subject batteries do not depend on scarce or geographically concentrated resources. Moreover, they offer promising safety benefits, because they are less susceptible to thermal runaway. They are, however prone, to shorter cycling lives.
Sodium–zinc molten salt batteries are attracting increasing attention as a viable solution, for managing intermittent renewable energies such as wind and solar. If we can resolve their relatively short cycling lives, we can expect them to play a useful role in the transition to renewable energy.
More Information
Battery Life Cycle vs. Cycle Life