Electrolytic capacitors have polarity, unlike ceramic and film ones that cannot handle direct current. Their positive anodes comprise aluminum, tantalum, or niobium metal. An insulating anodized oxide layer attaches to them to form the dielectric. Finally, a solid or liquid electrolyte encloses them as the negative anode

High Capacitance Effects of Electrolytic Capacitors
‘Capacitance’ is just another way to describe ability to store an electric charge. It avoids confusion because we use ‘capacity’ in many different contexts. Electrolytic capacitors have relatively high capacitance owing to the thinness of the anodized oxide dielectric. Furthermore, they have substantial anodes.
This large capacitance makes them ideal for storing bigger amounts of energy. In fact they can bypass or pass signals of several hertz. However reverse-polarity voltages, or ripple currents as low as a single volt can destroy the dielectric. This may result in catastrophic explosions, and fire.
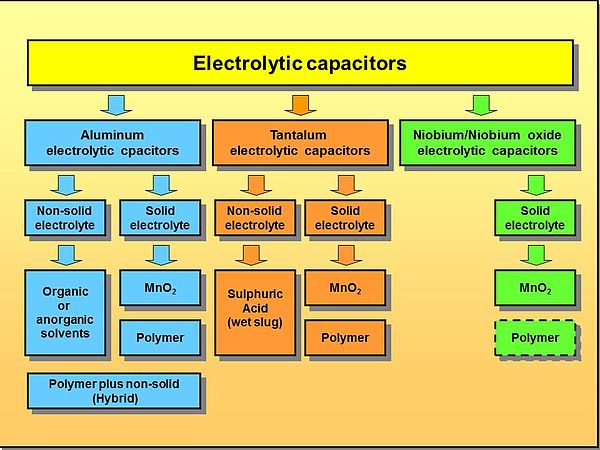
Varieties of the 3 Different Kinds of Electrolytic Capacitors
Aluminum versions use aluminum oxide as dielectric, while the others use tantalum and niobium pentoxides respectively. These surfaces are sintered or etched to have a rough surface structure. This creates a larger working area than a smooth, flat surface. The application process is similar to electroplating. The longer the voltage is applied, the thicker the layer but the lower the capacitance.
How Electrolytic Capacitors May Catch Fire and Explode
Because of the nature of the electrolyte, the positive terminal must always be at a higher voltage. If the voltage polarity reverses, the insulating dielectric may begin to fail resulting in a short circuit across the terminals. The large current running through it overheats the capacitor to a point where the enclosure bursts. This is similar to the process that may have caused Samsung Galaxy batteries to explode.
Related