A Case Western Reserve University media release dated December 12, 2024, that we somehow missed previously, caught our eye. The document posed the question why are lithium-ion batteries still so popular? When aqueous zinc-sulfur chemistry could do the same job at less cost, and with greater environmental sustainability. We thought we already had the answer, until we finished reading the research report.
Fixing The Downside of Aqueous Zinc-Sulfur Chemistry
Chase Cao is principal investigator, and assistant professor of mechanical and aerospace engineering at Case School of Engineering. His team announced significant progress in resolving aqueous zinc-sulfur chemistry limitations, that have been keeping this option away from consumer markets.
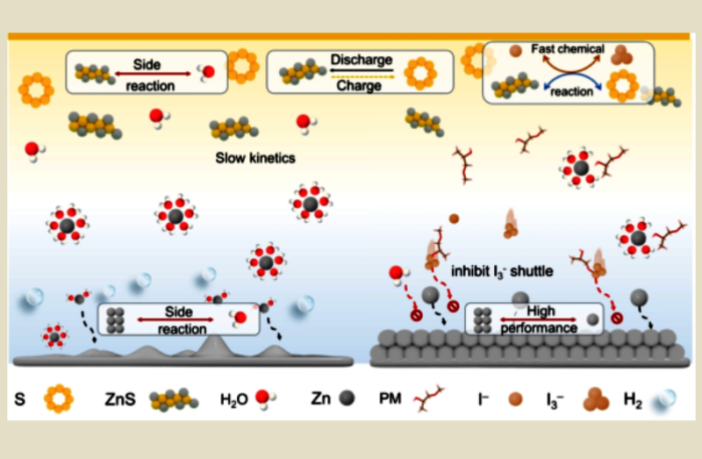
The challenges to overcome this time, are zinc-anode corrosion, low conductivity, and dendrite growth according to the media release. But Chase Cao and his team overcame all of these, by introducing propylene glycol methyl ether and zinc-iodide additives.
“This research marks a major step forward in the development of safer and more sustainable energy storage solutions,” the principal investigator explains.
“Aqueous zinc-sulfur batteries offer the potential to power a wide range of applications. From renewable energy systems to portable electronics, with reduced environmental impact and reliance on scarce materials.”
Towards a New Generation of Rechargeable Zinc Batteries
Chase Cao and his team achieved several crucial steps forward with their propylene glycol methyl ether and zinc-iodide additives:
- They enhanced energy capacity by 20%, while improving conductivity.
- They also improved stability, and inhibited the growth of zinc dendrites.
Dendrites and battery instability are the nemesis of many alternatives to lithium-ion. If these chemical growths worm their way past the electrolyte, then they can short-circuit the electrode charge carriers, and destroy the battery completely.
Guiyin Xu is a professor at Donghua University in Shanghai, and co-senior author of the research report. “These additives not only enhance battery efficiency,” he remarks. “They also address long-standing safety concerns by mitigating dendrite formation.
“The result is a compact, higher-density aqueous zinc-sulfur battery, that can recharge more times without significant degradation.”
More Information
Aqueous Zinc-ion Batteries Back on Table
Zinc And Air Batteries Could Outwit Lithium
Preview Image: Engineering Electrolyte Network Structure