In times gone past we popped a battery in our flashlight without a care in the world. However, perhaps it is time we understood batteries better, as they play an increasingly important role in our lives. We describe standard battery operating principles in this post, to help you get best value from your purchases.
Main Components Behind Battery Operating Principles
A typical battery cell has a metal anode able to give up electrons. This has a higher potential than its metal oxide or sulfide cathode. That cathode is able to receive those electrons, but first it requires a trigger. The stimulus occurs when the user inserts the battery in an open circuit, and the current begins to flow.
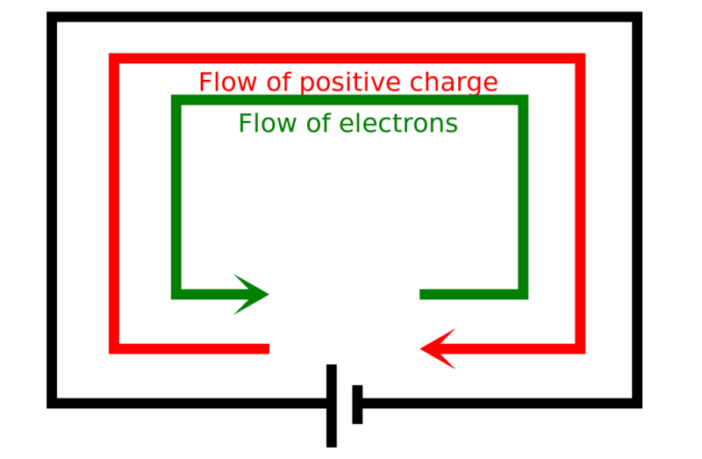
One battery terminal attaches to the cathode, and the other to the anode. Both of these internal components are surrounded by an electrolyte that controls the flow of the electrons between them. The metal that releases more electrons gains a positive charge, compared to the other metal that becomes negative in terms of standard battery operating principles.
The electrons flow through the external circuit from the positive to the negative battery terminal. This restores the balance in the battery. The battery becomes flat when all the electrons have moved across. However, a rechargeable battery can be restored close to its original state, by connecting it to an external electricity supply to replenish it.
Steps to Achieving Useful Battery Performance
A battery is only useful when it meets its user’s requirements in terms of energy capacity, and delivers this power at the desired rate. Those two factors depend in turn on the materials in the electrodes, and the performance characteristics of the electrolyte. Battery makers focus on ‘tweaking’ these components to optimize their performance, while scientists search for fresh ideas.
More Information
Our World of Batteries Looking In
Maintaining a Sealed Lead-Acid Battery