People visit our blog for a variety of reasons, and all are most welcome. We try our best to accommodate all their interests, and so our posts range across the full spectrum. We’ll be getting back to battery basics for the next couple of posts though. Returning to our roots, if you like, and reminding ourselves of the basic principles behind our products.
Back to the Basics – What Is a Battery?
A battery can mean several things in the English vocabulary. However, in our industry the term means an electro-chemical cell, that converts the energy in its chemicals into electric power. This definition contrasts with other devices (like solar panels) that convert other forms of energy into electrical energy.
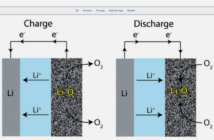
Getting back to battery basics, every electro-chemical cell has a positive, and a negative terminal electrode showing through the case. An electrolyte substance surrounds these terminals inside the case, to separate them so they cannot touch each other. However, the electrolyte does allow their net electrical charges to pass between them.
These electrical charges exchange between the terminals when a battery delivers its electricity. However, there are only so many of these ions available, depending on the chemical composition of the terminals. The battery becomes ‘flat’ after it has used up all of these. But this may not be the end of the road for device.
Two Types of Batteries Within the Same Design
There are two generic types of batteries. Understanding the differences could be your take away from the first episode of getting back to battery basics:
- The first generic type, namely primary or single-use batteries, are no further use after they have used up their ions. Although their materials should go to a recycling depot for reprocessing.
- The second generic type, secondary or multi-use batteries, can reinstate their charge when a user sends an external current through them, that returns the ions back to the original terminal.
This recharging process can keep a secondary battery going through many cycles. However, a few of the ions are permanently consumed each time. And so, eventually, the secondary battery should also go to a recycling depot for reprocessing.
More Information
Back to Battery Basics and Fundamentals