One of the less-efficient aspects of rechargeable batteries, is their chemistry is a two-edged sword. That’s because it also enables them to automatically reverse the process, and shed some of their energy over time. We discuss the phenomenon of battery self-discharge in this post. How serious is it and does it really matter?
How and Why Does Battery Self-Discharge Happen?
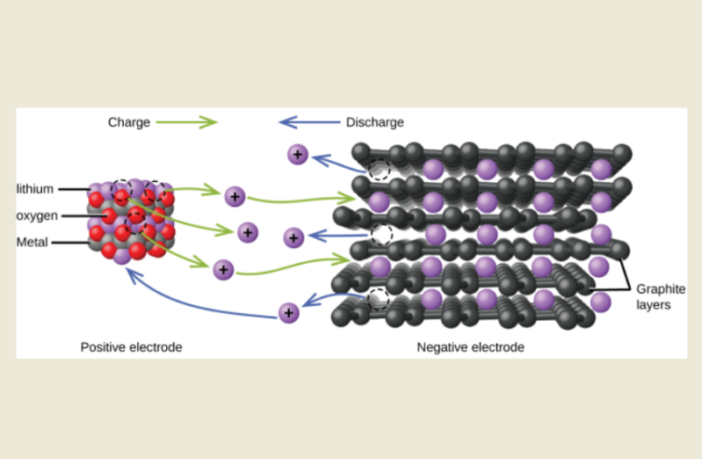
Rechargeable batteries have a particular chemistry, allowing their electrons and ions to return to their anode and electrolyte respectively. However, this unfortunately also opens the door to autonomous chemical reactions inside a charged battery, gradually draining it.
This autonomous discharge reduces a battery’s useful life between recharging cycles. It could even make them die completely, when left unattended in long-term storage.
- Lithium-ion rechargeable batteries may lose 2% to 3% a MONTH
- Lead acid rechargeable batteries may self-discharge at DOUBLE that rate
- Nickel-based rechargeable batteries could shed 10% to 15% per MONTH
Compare that to ‘old faithful’. Single-use alkaline batteries only use 2% to 3% a YEAR.
How to Control and Limit Self-Discharge Rate
We can influence battery self-discharge rates by behaving sensibly. However, the answer may not be as simple as we may think. The rate and intensity of self-discharge varies according to the ambient temperature. So it increases on hot days like many other chemical reactions.
But, if we are thinking ‘pop it in the refrigerator’ we need to think again. That’s because Panasonic confirms the moist air encourages self-discharge too, and could even damage the battery terminally.
The solution is quite simple when we think about it logically. Storage batteries like a cool, dry environment. Keep your batteries between 10 and 25°C for the best compromise. They won’t stop discharging. But this will slow the rate down, without damaging them internally.
More Information
Battery Memory Effect Is User-Driven