A lead-acid battery stores electrical energy, but in the form of chemical energy it converts back to the original version. It receives this energy from an external electrical source, but stores it chemically until required in the original form. Today we discuss some interesting aspects of charging and discharging lead-acid batteries. We hope you find this post interesting, and return to visit us again.
Some Technical Snippets about Lead-Acid Batteries
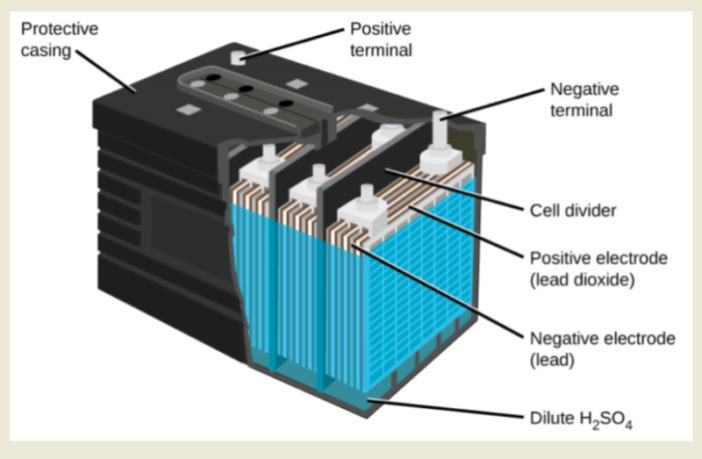
The lead-acid batteries that we, and other suppliers manufacture, still broadly follow Gaston Plante’s original 1859 design. This makes them arguably one of the most successful innovations of the industrial revolution, and there’s no sign of demand fading. All lead-acid batteries are capable of charging and discharging using these same constituents:
- A negative anode electrode comprising spongy or porous lead.
- A positive cathode electrode consisting of pure lead-oxide.
- Chemically permeable membrane separating the two electrodes
- A dilute sulfuric-acid solution serving as the electrolyte.
The nominal voltage of an individual lead-acid battery cell is 2 volts. Its total voltage is therefore a multiple of the number of its cells. However, the current capacity depends on the details of the design. For example, a 12-volt starter battery may be 4 amp hours, but an inverter battery could be as much as 150 amp hours.
More about Discharging and Charging Lead-Acid Batteries
ONE: DISCHARGING LEAD-ACID BATTERIES
A lead-acid battery in good condition begins to discharge smoothly the moment a user connects it to a matched load. Lead-sulfate crystals respond by drawing sulfate from the electrolyte, and forming on both electrodes. The battery stops discharging when the ions and electrons can no longer penetrate those crystals, and the sulfuric-acid solution becomes mostly water.
Two: RECHARGING LEAD-ACID CHEMISTRY
Recharging lead-acid cells reverses this process we described above. The lead-sulfate crystals decompose, and return the electrolyte and electrodes to almost their original state.
However, the cells weaken progressively until one no longer has capacity to function correctly. A technician then tells us ‘you have a dead cell’. It is time to purchase a new battery, and recycle the materials in the old one, so the process can begin all over again.
More Information
Pirate Lithium-ion Battery Ban in New York