India obtained local regulatory approval for a new DNA-based COVID shot, according to Bloomberg on August 20, 2021. Having primary local control should therefore put the world’s second largest population in a position to roll out vaccines faster. This DNA medication is the first of such products for the coronavirus out the starter gate. So how does this COVID DNA vaccine work, that’s apparently offering India fresh hope?
Let’s Learn the Basics of This Innovation
Vaccination stimulates the immune system with a harmless shadow of an infection, so it can learn how to cope with it. This then primes it to be ready when an actual infection occurs. Two different approaches have dominated the vaccination industry for a century according to World Health Organization. These are:
1… Introducing specific antigens against which the immune system reacts directly.
OR
2… Introducing live, but weakened infectious agents that replicate within the host.
It’s worth remembering that neither of these two approaches actually causes the disease.
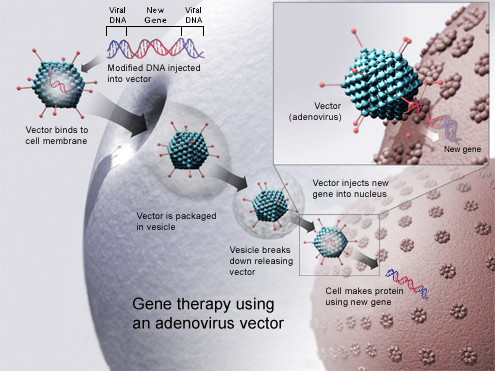
However, a COVID DNA Vaccine uses a different approach. That’s because it transfers the actual, specific antigen-coding DNA sequence into host cells. In simple terms, this means there is nothing for the immune system to learn or remember. The cells can start producing the antigen triggering the immune response right away.
Rolling Out the COVID DNA Vaccine in India
The vaccine is a three-dose preparation developed by Cadila Healthcare Ltd in Ahmedabad, India. It was 67% effective against symptomatic COVID in clinical trials in July, 2021. Moreover, Cadila claims it is equally effective against Delta and the other newer strains. Rolling it out should therefore help reduce hospitalizations and deaths in India from the virus. These have reached 32.3 million and almost 434,000 respectively.
The vast nation has previously largely relied on Astra-Zeneca and Pfizer vaccines which have been in short supply. Sputnik has only had limited use, while Johnson and Moderna are on hold pending agreement on legal indemnity. Cadila assures its product is suitable for children and adults 12 years and above. It hopes to produce 100-120 million doses annually, and is working on a two-dose version.
Recent
Dancing with Colds and Flu and Now COVID
Most Americans May Get Booster Shots
Preview Image: DNA Vaccine and Gene Therapy