Polymer electrolytes in all-solid-state lithium batteries transport ions, at a rate similar to liquid ionic solutions. This is a critical parameter in electrochemical batteries that influences their performance. A new study reveals that electrically conductive polymers offer advanced conductivity in solid-state lithium batteries. This could be a breakthrough.
More About Electrically Conductive Polymers
Electrically conductive polymers are a new trend in the history of batteries. Their conductivity may vary across a considerable range. But they also have flexible ion-transport capability, and variable junction and electrode effects depending on their physical properties.
This phenomenon opens up exciting potential attracting considerable research interest. There are great opportunities to manipulate this polymer’s chemical properties. This could lead to better solid-state lithium batteries, with all the advantages this brings.
Applying This Theory to Solid-State Lithium Batteries
All-solid-state lithium batteries continue to attract considerable interest, as an alternative to flammable lithium-ion alternatives. This attraction is due to solid’s potential high energy density, higher performance, and inherent safety characteristics for advanced energy storage systems.
These traditionally solid-state ceramic electrolytes also have high electrochemical stability, as well as high ionic conductivity to shuttle their ions. But on the downside, they are fragile, brittle, and their electrode-electrolyte interfaces are far from ideal.
A team from the Department of Mechanical Engineering, South Dakota School of Mines and Technology, has put this theory to the test. They published their findings in an MDPI Open Access Journal to which we link below:
- Traditional solid-state ceramic electrolytes (SSCE) require high cell-stack pressure. This can reduce the energy density of overall battery packages.
- Polymer-SSCE alternatives have inherently good inter-facial contacts with their electrodes. And they do not require high cell-stack pressures either.
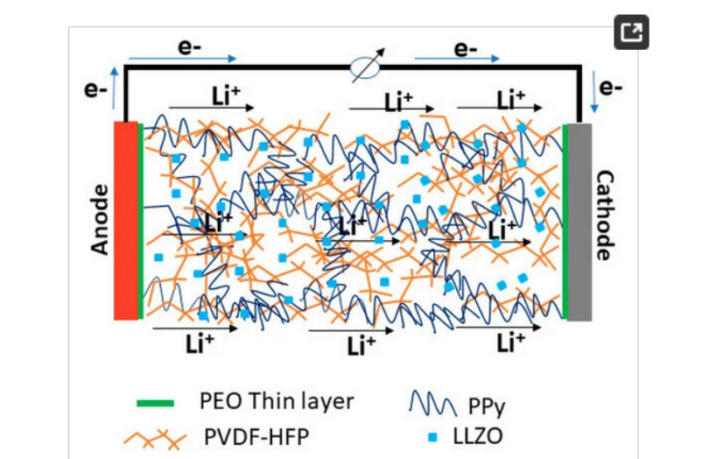
Electrically Conducting Polymers In a Polymer Backbone
The South Dakota School of Mines and Technology team incorporated a particular electronically and ionically-conducting polymer, to improve the performance of their polymer-SSCE model:
- This version demonstrated superior room temperature ionic conductivity, and electrochemical performance.
- It also displayed a high resilience to high temperature operation, compared to the liquid-electrolyte counterpart.
The team concluded that their electrically conductive polymers boost all-solid-state lithium battery performance. And that their novel approach might make a positive contribution to the next generation of these promising batteries.
More Information
Solid State Sulfide Electrolyte ‘Breakthrough’
Solid State EV Battery Race Accelerating