We round off our short series of articles about electro-chemistry, by reviewing common electro-chemical applications in our modern world. The four we mention below are practical applications that benefit society directly. Others may contribute to scientific experiments expanding our knowledge of our world, and our universe. This insight is expanding at a rate faster than ever before.
Exploring Four Primary Electro-Chemical Applications
Electro-chemistry, in a broad sense, involves the study of electricity, and how it relates to chemical reactions. We already know from our two previous posts that movements of ions between elements can generate electricity. But there is more to electro-chemistry than ‘just batteries’ as we shall see.
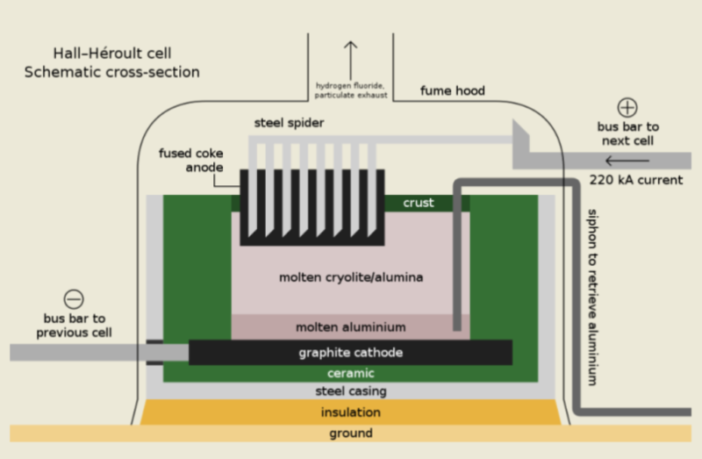
Electro-Chemistry in Metallurgy
Metallurgy concerns the properties of metals, and their production and refining. The mining industry sources, and purifies all major metals electro-chemically, with the exception of iron and steel.
Electro-Plating Common Metals
The electro-plating process, or electro-chemical deposition, deposits one metal over another in the presence of a water-based solution of metal salt. This is a popular way of decorating base metals, and resisting corrosion according to Britannica.
Electrolysis of Brine
Electrolysis of brine, or a strong solution of salty-water, is the third of the four electro-chemical applications we highlight today. Producing chlorine and caustic soda is one example, but there are others too.
Electro-Chemical Applications in Energy Storage
Super capacitors, and storage batteries have become key technologies in industrial applications. However, more recently the focus has shifted to electric vehicles, and load-leveling in the electricity utility industry.
Both applications have become fundamental building blocks in the shift away from oil-based fuel. Electro–chemical batteries and fuel cells are spearheading major efforts to halt global warming, and navigate away from climate change too.
More Information
Electro-Chemical Reactions Unpacked For You
Electrolytes in Electro-Chemistry Moving Ions
Preview Image: Hall–Héroult Aluminum Electrolysis