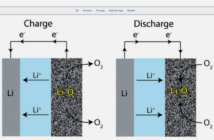
What use could energy in batteries be, unless we could convert it to something valuable we could use? Science describes this remarkable property as being able to do work. That work, we assume, must be something that adds value to our lives. In the case of electrochemical batteries, that something is electricity.
Energy in Batteries Assumes Many Forms
Energy in batteries can assume many different forms in order to do useful work. The U.S Energy Information Administration (EIA) mentions the following, although we are confident there are more:
- Chemical energy to create electricity for our work.
- Heat energy to keep us warm and cook our food.
- Light energy so we can find our way in the dark.
- Motion energy to turn a motor to work a machine.
- Electrical energy to power this amazing website.
- Gravitational energy to lift a weighty object.
We never cease to be amazed by the potential of our humble batteries! Of course batteries are not the only store of energy. Timber contains energy too, which we release by burning it. Food contains energy to sustain us, and so on.
The Difference Between Potential and Actual Energy
Potential is a latent state, with the capacity to deliver given the right trigger. And so we might speak of a ball at the top of a hill, just requiring a tap to get it rolling. If it did, we would see actual energy in action, as gravity drew it down the slope.
Even so, on the same basis the electrochemical energy in a battery is only potential. We first have to create an external circuit between the terminals to transform it into actual, electrical energy. So simple, so elegant is electricity, wisely used in action.
More Information
Trickle Charging a Lead Battery for Health