Macrocycle-cage molecules have rings of twelve or more atoms each. These structures can be used as building blocks for lithium-ion battery solid-state electrolytes. Those electrolytes will then have extremely high conductivity. A pair of Cornell University scientists have fused these macrocycle-cage molecules together, to create porous crystal structures. These structures can take up lithium-ion electrolytes, and transport them smoothly via one-dimensional nano-channels.
Finding a Way Forward to Safer Lithium-Ion Batteries
The project was a joint venture between Yu Zhong, assistant professor of materials science and engineering at Cornell, and undergraduate transfer student Kaiyamg Wang. Zhong was keen to find a way forward to safer lithium-ion batteries.
Conventional lithium-ion batteries shuttle their ions through liquid electrolytes. But those liquid electrolytes can form spiky dendrite growths between their anodes and cathodes. This can in turn create catastrophic short circuits between two electrodes, destroying the battery and leading potentially to explosions.
Zhong and Wang agreed that solid-state batteries could avoid these situations. Although they were concerned that greater resistance could cause the ions to move more slowly through solid materials. Zhong realised they needed a new porous crystal to facilitate ion transfer.
That pathway would have to be smooth and without any strong interactions, the team knew. Or else the pathway might cause the lithium ions to become trapped. While at the same time, the crystal should be able to accommodate sufficient ions to form a high concentration.
Safer Lithium Batteries With Fused Macrocycle-Cage Molecules
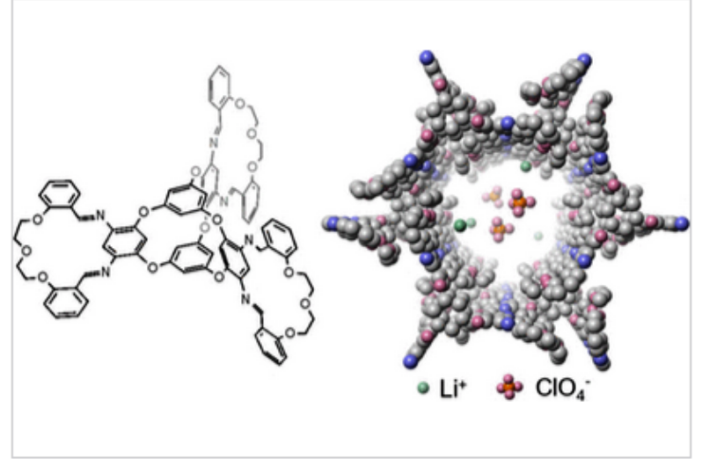
“Both macrocycles and molecular cages have intrinsic pores where ions can gather and pass through,” Wang explains. Therefore, he reasoned, if they combined the two elements this would create a crystal with large spaces to accommodate ions.
Zhong and Wang created multiple fused structures, each with three macrocycles radially around molecular cages at the center. These structures used hydrogen bonds to interlock with each other, and create three-dimensional crystals that were nanoporous with one-dimensional channels.
This measure formed “the ideal pathway for the ions to transport,” Zhong advises. “That conductivity is the record high for these molecule-based, solid-state lithium-ion-conducting electrolytes.”
Assembling the components in this form was previously an unexplored area, and so his team found their way forward to safer lithium batteries.
More Information
Reducing Resistance in Solid State Batteries
The Solid State Battery Game Changer