That’s a fair question, given that iron is cheap and abundant, whereas lithium and cobalt are quite rare and expensive. And moreover, iron does not catch fire when it overheats, and explodes. The worst iron does is melt at extremely high temperature, by which time that could be the least of our worries. Researchers at Oregon State University wondered is iron the key to better batteries, and decided to find out.
Reactivity Is the Key to Better Iron Batteries
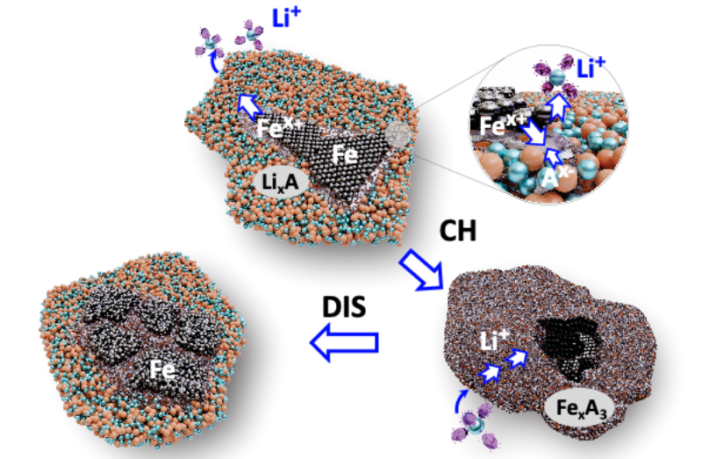
“We’ve transformed the reactivity of iron metal, the cheapest metal commodity,” Oregon State’s Xiulei “David” Ji explains. “Our electrode can offer a higher energy density than state-of-the-art cathode materials in electric vehicles.
“And the cost of our batteries is potentially much lower, since we use iron whose cost can be less than a dollar per kilogram. This is a small fraction of the price of nickel and cobalt, which are indispensable in current high-energy lithium-ion batteries.”
The study at Oregon State University was in conjunction with Argonne National Laboratory, Vanderbilt University, Stanford University, the University of Maryland, Lawrence Berkeley National Laboratory and SLAC National Accelerator Laboratory.
Integrating Iron Into the Normative Battery Structure
Batteries store their energy in chemicals, whose reactions convert this energy into the electricity we use to power our applications. There are many different varieties, but they all use cathode and anode electrodes. When we discharge a battery, its energy flows from the anode to the cathode via the application.
Meanwhile, the electrical charge is carried by lithium-ions in a lithium-ion battery. These move through the electrolyte from the cathode to the anode during discharging, and back again during charging. The Oregon State University’s iron battery will work in a similar way.
“Our iron-based cathode will not be limited by a shortage of resources,” a university spokesperson says. “Iron, in addition to being the most common element on Earth as measured by mass, is the fourth-most abundant element in the Earth’s crust.
“We will not run out of iron till the sun turns into a red giant. We’ve demonstrated that the materials design with anions can break the ceiling of energy density for batteries that are more sustainable and cost less.” So is iron the key to the future of batteries? We’ll sure add it to our list!
More Information
Nickel-Iron Battery History You May Not Know
An Iron Age of Batteries Could Be Here