A battery is a sealed device containing energy stored in chemicals, that it converts to electricity. The device may be a single module, or multiple cells in direct or parallel series. Stay with us and learn how battery chemistry works, as we expand these principles in straightforward terms.
Electricity and How Battery Chemistry Stores It
Electricity is a form of energy produced by a flow of ions. Ions are atoms, or groups of atoms that carry positive or negative electric charges, as a result of having lost or gained one or more electrons. Please stay with us for a while longer, to learn how battery chemistry works in more detail.
Electrons in turn are stable subatomic particles with negative electric charges. We find these particles in all atoms, and they are the primary carriers of electricity in solids. Electrons materialize in a battery through a chemical reaction at one electrode, and then flow over to the other electrode.
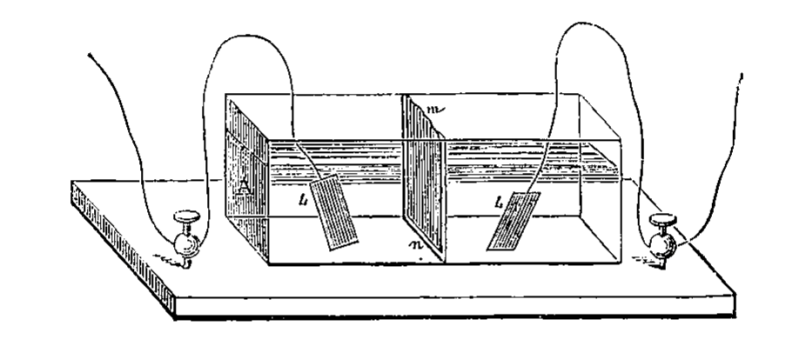
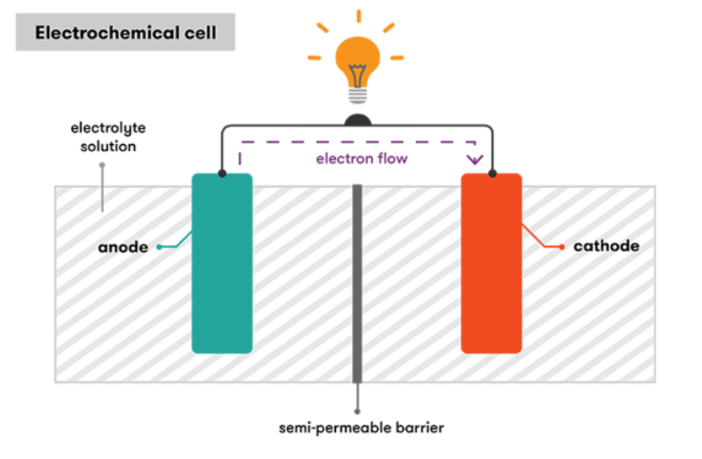
The electrodes at ‘L’ in the above diagram, are the source and the destination of the electrons respectively. We call the source electrode the ‘anode’, and the destination electrode the cathode. These two components are usually different metals, or chemical compounds.
Fair Enough, But Where Does the Anode Get Its Electrons?
The anode in the drawing reacts with the electrolyte, also containing the cathode on the far side of the separator. This reaction produces the electrons that accumulate at the anode. Meanwhile, a second, different reaction occurs simultaneously at the cathode, preparing it to receive the electrons.
The presence of the neutral electrons at the anode produces a matching amount of positively charged ions at the cathode, to maintain a neutral charge balance.
A third chemical reaction sends the electrons from the anode to the cathode through an external circuit, causing the electricity that powers the device. Meanwhile, the ions travel from the cathode to the anode, to restore the electro-chemical balance in the battery.
More Information
Battery Fundamentals and Flow of Electrons