A battery converts the chemical energy inside its case to electricity, but only when we connect it to an outside circuit. That circuit could be your phone, for example, or your watch. You’ll find more information by clicking on the basic tab at the top of this page. Or you could stay on this page, and learn more about electric batteries right here.
The Basics Inside Every Electric Battery
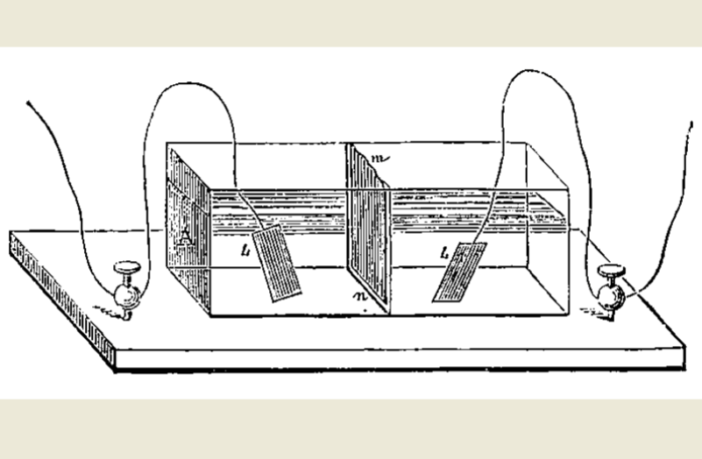
A battery has a positive and a negative ‘terminal’ on its case, for connecting to an external circuit. Those two connection points lead to corresponding positive and negative plates inside the battery. We illustrate this principle in the drawing at the top of this article.
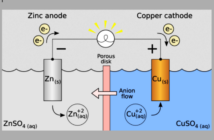
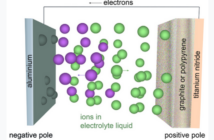
Electricity leaves the battery from the negative anode plate. And then completes the loop through the circuit before returning to the positive cathode in the battery. But there is more about electric batteries to know that just that.
Because, if the electricity were to flow directly between the terminals inside the battery, there would be a short-circuit releasing all the energy in an instant. This would generate a large amount of heat, which is the source of lithium-ion battery fires.
Battery makers insert separator plates between the electrodes, to prevent them touching directly. However, these plates are selectively perforated, to allow ions to flow through electrolyte in the opposite direction while the battery is powering the device. When all the ions have made their journey the battery is ‘flat’.
Types of Batteries For Different Purposes
Batteries come in different sizes and shapes to suit their purpose. Any one of them is ‘flat’ when all the ions have moved from the cathode to the anode. In some batteries we can reverse this flow by sending electricity through the battery, in the opposite direction from which it left.
This recharges the battery so it is ‘full’ again, not ‘flat’. We call batteries that can do this, ’secondary’ batteries, because we can use them a second, and many more times. However, not all batteries can do so. When ‘primary’ single use batteries are ‘flat’, they are due for recycling.
More Information
Battery Electrode Roles – How They Flip