Okay, so you went on a school expedition where they showed you a Leclanche cell? Maybe they went as far as explaining you were looking at a very early battery. Although perhaps it looked more like a cookie jar to you. Actually, it contained those same familiar elements of cathode, anode, electrolyte, terminals and case. Let’s take it apart in our minds, and figure out what once happened inside.
The Secret Life of a Ground Breaking Leclanche Cell
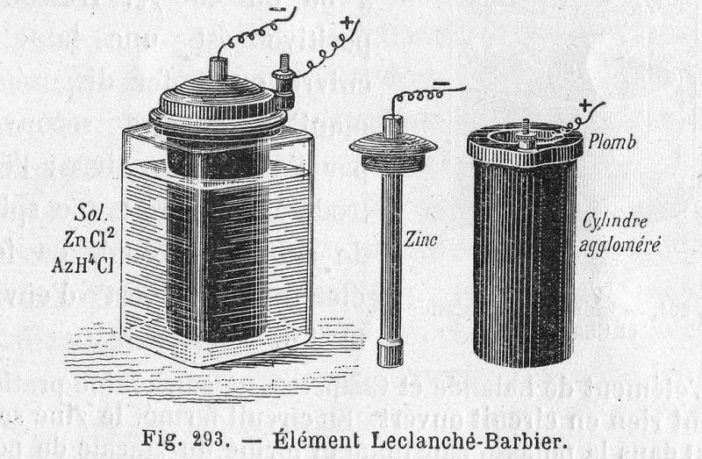
The revolutionary battery cell George Leclanche cobbled together in 1865 was an unglazed clay pot. This contained a mixture of powdered carbon and manganese dioxide, plus a carbon rod standing upright in the middle.
The unglazed pot with its contents stood inside a glass jar containing a saturated ammonium chloride solution. Plus a zinc rod standing upright in the available space. The carbon and zinc rods were the two electrodes, while the solution was the electrolyte. The opening at the top of the jar had a waterproof cap to prevent the contents leaking.
What Would Happen If We Charged the Cell?
We can’t promise the Leclanche cells in museums are still working. Perhaps it’s better to leave them alone. However, this is what should have happened when the inventor applied external power:
- The electrolyte split into ammonium and chloride ions. The chloride ones moved towards the zinc rod, and surrendered their electrons forming zinc chloride. The zinc rod became the negative cathode as a result of the electron excess.
- Meanwhile the ammonium ions moved to the carbon rod, where the free ions there neutralized them and formed ammonia and hydrogen gas. The carbon rod the became the positive anode due to a deficiency of ions.
The manganese dioxide oxidized the hydrogen gas, to form water. Approximately 1.5 volts of electricity flowed from the cathode to the anode, after Leclanche added an external circuit. This may seem commonplace nowadays. However back in those olden days, this was early rocket science!
More Information
George Mansbridge Metalized Paper Capacitor
Charles Pollak Aluminum Electrolytic Capacitor