Scientists at Brock University in Ontario, Canada, have developed a new substance that could increase battery efficiency. Their solid polymer electrolyte material (SPE), could be just what our industry needs for next generation battery separators.
Future of SPE Solid Polymer Electrolytes
Solid polymer electrolytes are promising materials for next-generation solid-state lithium-ion batteries. This is on account of their mechanical flexibility, ease of assembly, and greater safety.
However, SPEs inability to conduct ions as well as liquid electrolytes is holding them back. The Brock University team successfully tackled this problem in a revolutionary way, offering a promising pathway forward.
The Brock solution lies in using a novel solid polymer electrolyte in the battery separator that keeps the electrodes apart, while still allowing the ions to flow. They chose their particular SPEs because they help protect the environment too. However, the team was still not satisfied with ion transfer speed.
Accelerating Ions in SPE Materials
The speed with which ions travel affects the rate at which batteries charge, and discharge. The researchers at Brock University did not find what they needed among existing commercial polymer membranes.
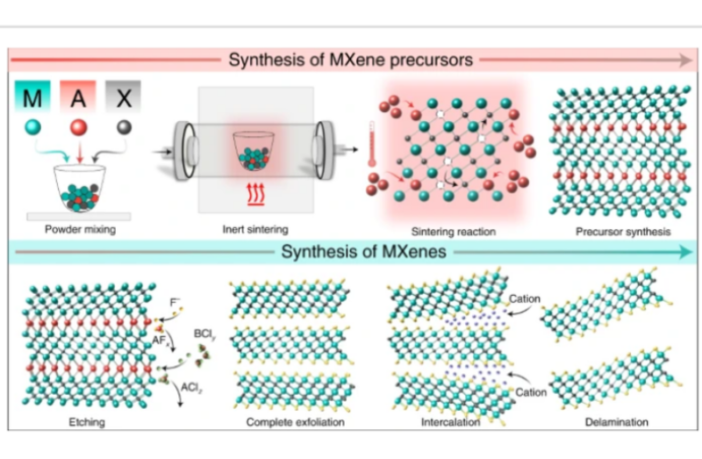
And so they synthesized an advanced material, titanium carbide MXene. Then they incorporated this material into various polymer matrices to create the solid polymer electrolyte material they needed.
Their membranes turned out a treat! They were stronger than standard ones, and enabled more effective ion movement between terminals. In fact, the researchers described them as ‘acting like highways’.
Durable Polymer Electrolytes For Solid State Batteries
The researchers received an added bonus for their work. Their advanced titanium carbide MXene turned out to be stronger than conventional polymer materials. This makes their solution a great candidate for next generation solid state lithium-ion batteries.
“Conventional membranes start to degrade at higher temperatures,” they told Education News Canada. “In contrast, our membrane remains stable under high-temperature and high-humidity conditions.”
More Information
Biodegradable Battery With Natural Polymers
High Density Polymer Electrolyte Boosts L-Ion
Preview Image: Top Down Etching Routes