When comparing (all) solid state batteries, we include both solid-state battery and all-solid-state battery types. Folk often use these terms as if they mean the same thing. The truth is there a differences between them, and these differences matter.
The Differences Between (All) Solid State Batteries
We have to separate the two types, when meaningfully comparing (all) solid state batteries. In a nutshell, solid-state batteries set the stage, while all-solid-state ones improve the design.
Solid-State Batteries (SSB)
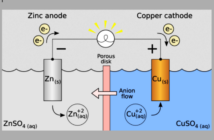
A solid-state battery replaces traditional liquid electrolyte with a solid material, although this still carries the charge between the electrodes. This solid electrolyte might be a ceramic, glass, sulfide, or polymer material.
However, and this is important, many solid-state batteries still contain some liquid or gel components. These may be inside their electrodes, or at the interfaces between layers.
In practice then, SSBs are often hybrid designs aiming to improve safety by reducing flammable liquids. While at the same time they still use small amounts of soft materials, to maintain electrical contact between parts of the battery.
All-Solid-State Batteries (ASSB)
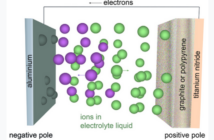
The all-solid-state battery design goes further. Every internal component is solid. This includes the electrolyte, the separator, and even the contact layers between the electrodes.
There are no liquid or gel materials anywhere inside the cell. This fully solid architecture brings several potential advantages:
- The fire risk greatly reduces in the absence of flammable liquids.
- The storage capacity increases, meaning more power in less space.
- The battery can recharge and discharge itself many more times.
The battery is also more thermally stable, even with lithium metal anodes. However, all-solid-state batteries are much harder to manufacture. All-solid-state battery interfaces tend to develop microscopic gaps, as materials expand and contract during charging.
These gaps increase resistance, and reduce performance over time. Engineers therefore have to tackle issues such as brittleness, interfacial contact loss, and dendrite formation, without relying on liquids to ‘self-heal’ the structure.
More Information
Copper in Solid-State Sulfide Batteries