Mahatma Gandhi University in Kerala, India, has extensive experience in electrode research. It’s current themes include developing flexible electrodes, and improving the performance of batteries, fuel cells, and supercapacitors. The University recently published a research paper describing a procedure for upcycling L-ion for supercapacitor electrodes.
Breaking Down Lithium-Ion Batteries for Upcycling
The increasing popularity of electric vehicles and portable electronic devices is leading to a growing pile of battery waste. Upcycling this waste directly to other applications can be more beneficial than recycling it completely.
The Mahatma Gandhi researchers applied this theory to upcycling selected lithium-ion (L-ion) battery materials for re-use as high-performance supercapacitor electrodes. This signals a potential new road ahead.
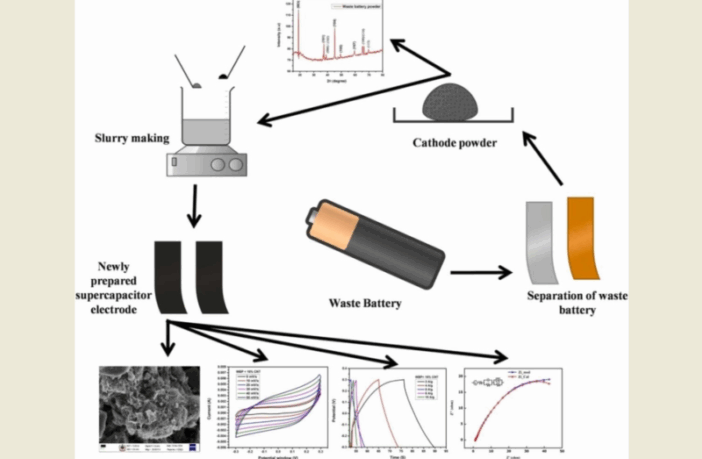
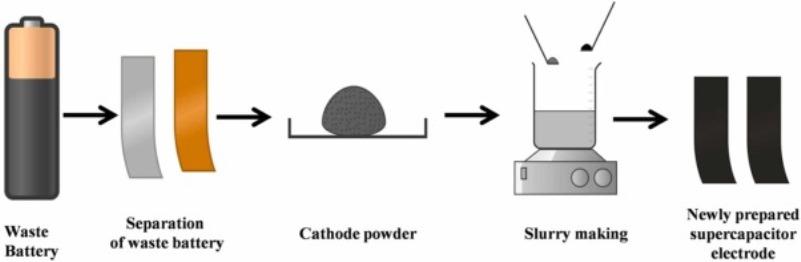
The diagram at the top of this page provides a handy overview of the upcycling process, per the research report we link to below:
- First, the researchers sourced a stock of spent lithium-ion (L-ion) batteries in their local area.
- Before proceeding further, they soaked these batteries in a mild sodium chlorine solution.
- This step deactivated the spent lithium-ion batteries, so they were safe to take apart.
- The team then rinsed the batteries in de-ionozed water, to remove any surface contamination.
- Finally, they thoroughly dried the batteries in an oven at 60° C / 140° F for 10 hours.
This was the final step in preparing the batteries for upcycling their L-ion cathodes for supercapacitor electrodes. The researchers then dismantled the cleansed batteries to separate their cathodes, anodes, and separators. They were particularly interested in recovering the cathode materials and proceeded as follows:
- They soaked the cathodes in dimethylformamide solution to loosen lithium-based oxides on the aluminum current collectors.
- Next, they subjected the cathodes to ultrasonic treatment for two hours to detach those oxides.
- Finally, they heated the purified cathodes for five hours in an oven, that stepped up to 700° C / 1,292° F.
The Mahatma Gandhi University researchers then ground the cathodes into fine power, mixed with a carbon nanotube material for structural strength. They converted this compound to slurry by adding a binder. Then they coated the result onto copper foil for drying, in the final stage of upcycling L-ion cathodes for supercapacitor electrodes.
More Information
Upcycling Instead of Recycling Batteries
Supercapacitors and Batteries Head to Head