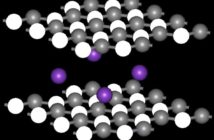
It is always a bit difficult to explain chemical equation balancing to students initially. As with everything else, the first few classes decide whether they will develop interest in the new topic or not. So we are going to use candies to explain and demonstrate the concept of balancing chemical equations. As candy is something all kids can relate to, it will be great fun, whether done in class or at home.
Materials Required
- 10 small pieces each of 7 Colors of candies
- Three A4 sized white paper
- Black color (ink) sketch pen.
Procedure
We will write and balance these equations using candies:
- Na + Cl2 = NaCl
- Na + H2O = NaOH + H2
- CO + NO = CO2 + N2
- Fe2O3 + CO = Fe + CO2
- 3C + Fe2O3 = 3CO + 2Fe
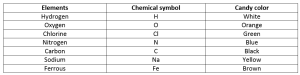
Make groups of two in your class. Associate candies, depending on their color, with specific atoms. A suggested association is given in the table in Figure 1. Each student should have a jar of candies as per the table.
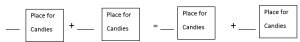
Next, draw box, blank space, + sign and = sign on chart paper with the help of sketch pen for equations. A drawing has been given below in Figure 2.
Now ask the students to put the number of candies on the paper as equation shows. Make one student responsible for each side (left and right) of the equation.
Next ask them to add candies such that number of candies on the left and right of = sign are same. For example, in the first equation, these steps have to be followed:
- There are 2 green candies on the left but only one on the right. So, 1 green candy has to be added on the right.
- Green candy is grouped with yellow candy on the right. So 1 yellow and 1 green candy have to be added on the right.
- Step 2 makes 1 yellow candy less on left. So 1 yellow candy has to be added on the left too.
- Step 3 makes number of yellow and green candies equal (2 each) on both the sides.
Similarly, all other equations have to be balanced, as shown in Figures 3-7.
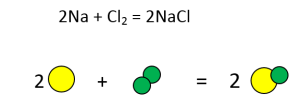
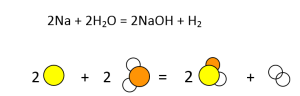
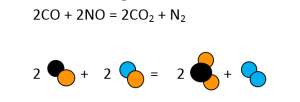
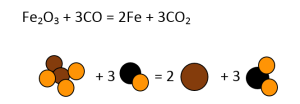
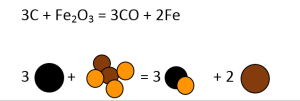
Conclusion
After doing the experiment outlined above, students will be able to easily understand the atomic structure of element and learn how to balance the chemical equations too. Care needs to be taken that the candies used for experiment are not consumed by the students during or after the experiment. However, those still left in the jar can be enjoyed as a reward for successful experimentation!!