Graphene aluminum-ion batteries are a fairly new technology, aiming to charge faster and last longer than lithium-ion. The University of Queensland in Australia researched the chemistry and confirmed that it showed promise. A joint project involving the private sector is progressing well. But first, how do these batteries work?
How Do Graphene Aluminum-Ion Batteries Work?
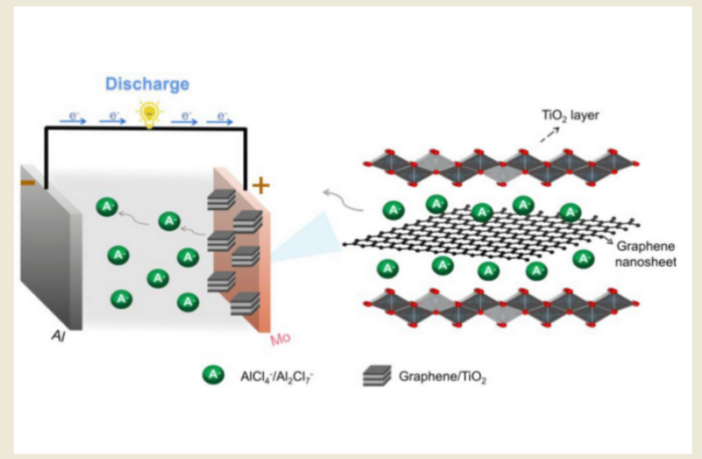
This revolutionary battery design uses aluminum in its anode, and graphene for its cathode. The electrode is typically a liquid containing aluminum chloride salts.
When the battery discharges, the aluminum atoms shed electrons and become aluminum ions. These electrons move through the external circuit, while the ions settle in the graphene cathode.
When the battery recharges, the above processes reverse out. The aluminum ions return to the aluminum anode, regain their lost electrons, and become aluminum atoms again.
But there is a catch to this seemingly ideal world. Each aluminum ion carries three charges. This makes the chemistry bulky, and reduces the amount of energy the battery can store per size and weight.
And so, lithium-ion batteries have held on to their lead in terms of energy capacity. Although we have heard that scientists are stretching the energy-carrying capacity of graphene aluminum-ion batteries.
Breaking News About These Batteries
The University of Queensland private sector partner is working in tandem with a major mineral mining company. They now report significant progress with their graphene aluminum-ion batteries.
In summary, this progress means considerably improved battery performance during six-minute charging cycles. This could fundamentally change prospects for electric vehicles, consumer electronics, and stationary storage too.
The enhanced graphene aluminum-ion battery also redoubles its energy-carrying capacity when fully charged. We understand that work is ongoing to further optimize the anode, the cathode, the electrolyte, and the overall cell design, and we will advise further progress.
More Information
Graphene Cathodes Enhance Conductivity
Aluminum-Ion Batteries Are Resurfacing