The chemistry in a battery is remarkably simple in theory, yet what makes it tick may seem a miracle to many. In summary terms batteries store chemical energy and release it as electricity. But we are running a little ahead of ourselves. Let’s return to the beginning of the story and start all over again.
How the Chemistry in a Battery Makes Electricity
Now electricity is a type of energy resulting from a flow of electrons. In case you wondered, electrons are subatomic particles in all atoms, and they hold negative electricity charges. However, for them to flow (and produce electricity) they need somewhere to flow from, and to.
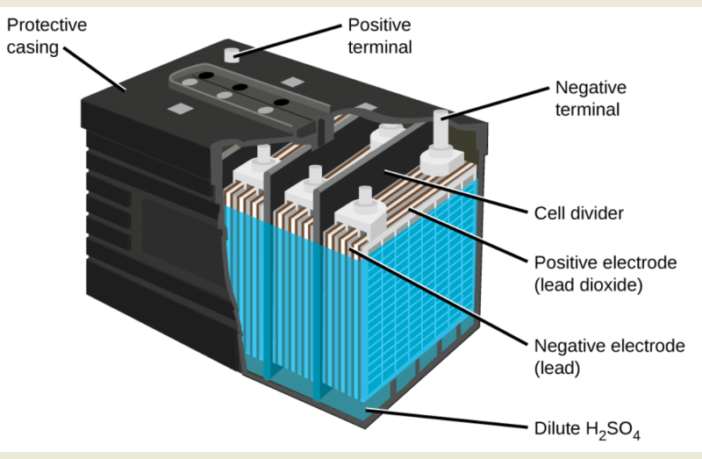
In batteries, we call these ‘arrival and departure terminals’ electrodes. The electrons depart from a negative one we call an anode, and flow across to a positive electrode we call a cathode. However, those two electrodes are almost always different materials, but complementary.
But it stands to reason we can’t have chaotic chemistry in a battery where the electrons flow in an uncontrolled fashion. A third component we call the electrolyte separates them. It acts as a gatekeeper controlling the flow of the electrons between them.
Where Do These Electrons Come From?
The negative anode electrode reacts with the electrolyte to produce and store electrons. Meanwhile, at the positive cathode a second chemical reaction prepares it to receive those electrons. Everything is in place to produce the electricity, but the battery is still in a neutral state. Alas we are running out of time again and we will have to continue in the next post.
The world of batteries is growing rapidly as the need for energy storage expands. Yet the same principles still apply in electric cars, smartphones, computers, and even space probes. Join us on our journey to explore the secret life of batteries you may not know.
Breaking News
Recycling Lithium-Ion Batteries With CO2